Chapter 2: Q 27 E (page 59)
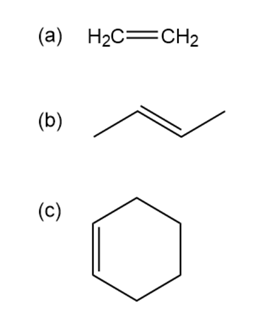
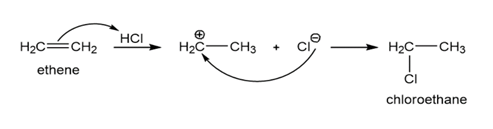
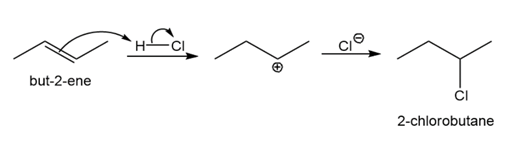
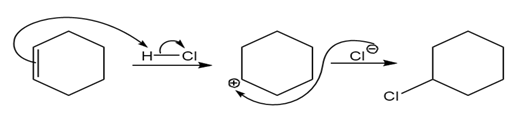
Double bonds can also act like Lewis bases, sharing their electrons with Lewis acids. Use curved arrows to show how each double bond below will react with HCl and draw the resulting carbocation.
Short Answer
a)
b)
c)
Learning Materials
Features
Discover
Chapter 2: Q 27 E (page 59)
Double bonds can also act like Lewis bases, sharing their electrons with Lewis acids. Use curved arrows to show how each double bond below will react with HCl and draw the resulting carbocation.

a)

b)

c)

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What is the Ka of HCN if its pKa=9.31?
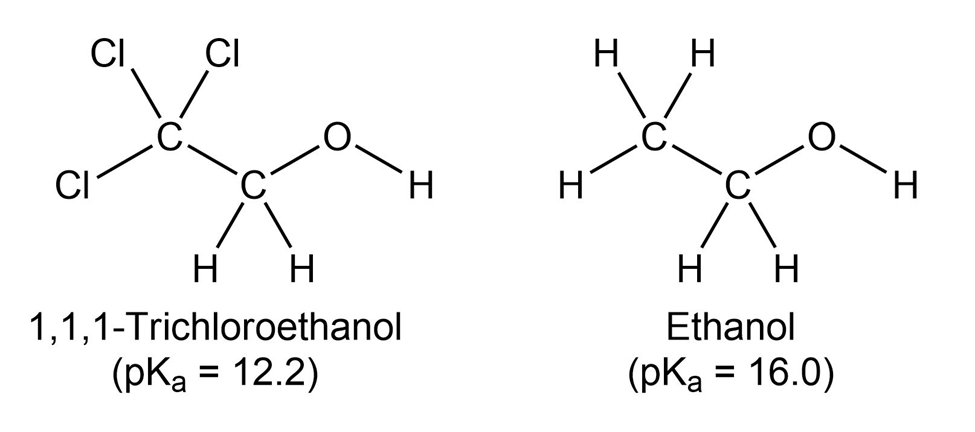
1,1,1-Trichloroethanol is an acid more than 1000 times stronger thanethanol, even though both have a conjugate base where the negativecharge is on an oxygen. Provide an explanation for this observation.
Question: Organic phosphate groups occur commonly in biological molecules. Calculate formal charges on the four O atoms in the methyl phosphate dianion.
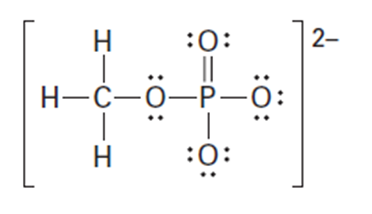
Methyl phosphate
Which intermolecular force is predominantly responsible for each
Observation below?
(a), a component found in paraffin wax, is a solid at
Room temperature while octane is a liquid.
(b) has a higher boiling point than
(c), which is found in vinegar, will dissolve in water but not
In oil—for simplicity you may assume oil is .
Will either of the following reactions take place to a significant extent as written, according to the data in Table 2-3?
a) b)
What do you think about this solution?
We value your feedback to improve our textbook solutions.