Chapter 2: Q.2-2-16P (page 47)
Question: What is the Ka of HCN if its pKa=9.31?
Short Answer
Answer
The Ka of HCN is if its pKa= 9.31.
Learning Materials
Features
Discover
Chapter 2: Q.2-2-16P (page 47)
Question: What is the Ka of HCN if its pKa=9.31?
Answer
The Ka of HCN is if its pKa= 9.31.
All the tools & learning materials you need for study success - in one app.
Get started for free
Rank the following substances in order of increasing acidity:
Draw the conjugate base for each compound below (the acidic hydrogen in each case is marked with an *).
(d)
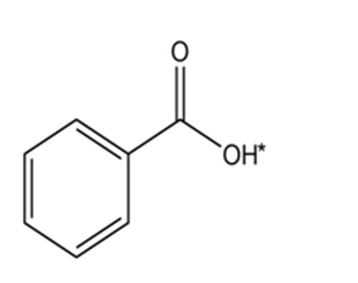
Use the electronegativity values shown in Figure 2-2 to rank the following bonds from least polar to most polar: H3C–Li, H3C–K, H3C–F, H3C–MgBr, H3��–O��
Question: Draw as many resonance structures as you can for the following
species:
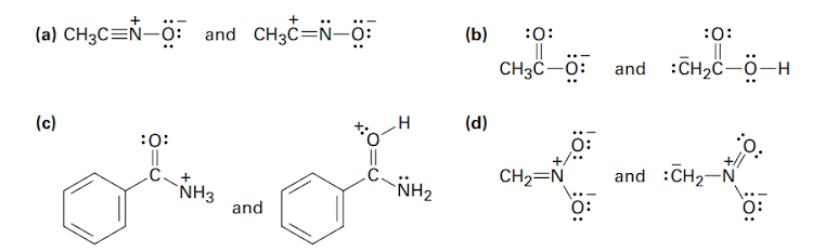
Which of the following pairs represent resonance structures?
What do you think about this solution?
We value your feedback to improve our textbook solutions.