Chapter 2: Q 26 E (page 59)
Question: Use the curved arrow formalism to show how the electrons flow in the resonance form on the left to give the one on the right.
a)
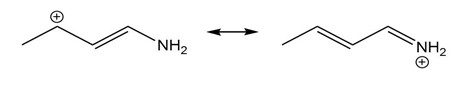
b)
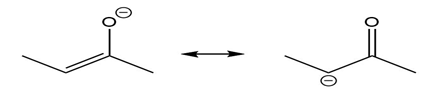
c)
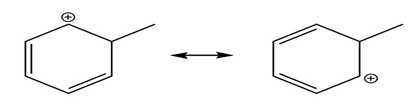
Short Answer
a)
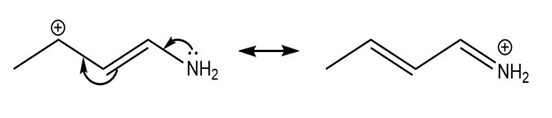
b)
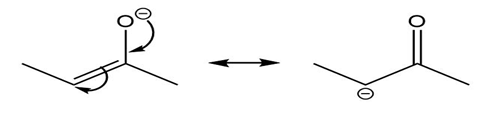
c)
Learning Materials
Features
Discover
Chapter 2: Q 26 E (page 59)
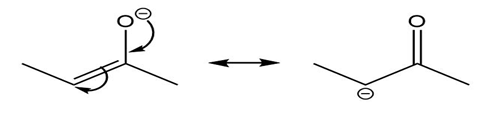
Question: Use the curved arrow formalism to show how the electrons flow in the resonance form on the left to give the one on the right.
a)

b)

c)

a)

b)

c)

All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the conjugate base for each compound below (the acidic hydrogen in each case is marked with an *).
(a)
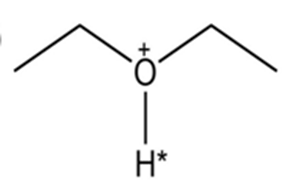
Predict the structure of the product formed in the reaction of the organic base pyridine with the organic acid acetic acid, and use curved arrowsto indicate the direction of electron flow.
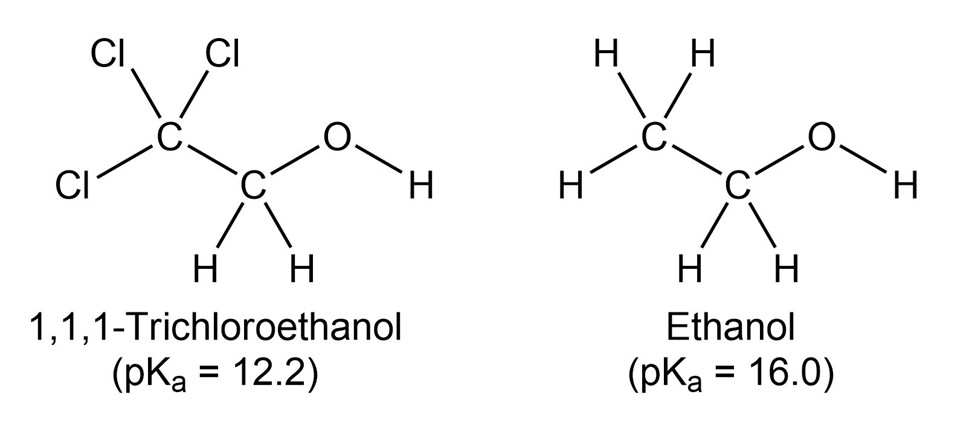
1,1,1-Trichloroethanol is an acid more than 1000 times stronger thanethanol, even though both have a conjugate base where the negativecharge is on an oxygen. Provide an explanation for this observation.
Determine if each compound or ion below has a dipole moment.
(a)Carbonate ion
Maleic acid has a dipole moment, but the closely related fumaric acid,
a substance involved in the citric acid cycle by which food molecules
are metabolized, does not. Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.