Chapter 2: 51E (page 59)
What is the pH of a 0.050 M solution of formic acid, =3.75?
Short Answer
The pH of a 0.050 M solution of formic acid is 2.52.
Learning Materials
Features
Discover
Chapter 2: 51E (page 59)
What is the pH of a 0.050 M solution of formic acid, =3.75?
The pH of a 0.050 M solution of formic acid is 2.52.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Which of the following pairs of structures represent resonance forms?
Use the electronegativity table given in Figure 2-2 to predict which bond in each of the following pairs is more polar, and indicate the direction of bond polarity for each compound.
a)or
b)or
c)or
d)or
Question: Calculate the formal charges on the atoms shown in red.
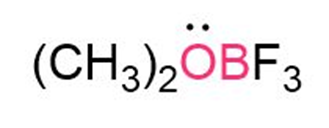
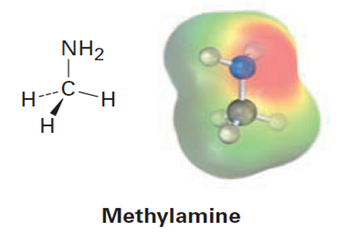
Look at the following electrostatic potential map of methylamine, a substance responsible for the odor of rotting fish, and tell the direction of polarization of the C-N bond:
The ammonium ion ( , pKa=9.25) has a lower pKa than the
methylammoniumion ( , pKa=10.66). Which is the stronger
base, ammonia or methylamine ? Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.