Chapter 2: Q.2-2-35_A (page 35)
Question: Calculate the formal charges on the atoms shown in red.
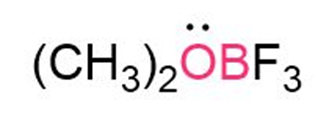
Short Answer
Answer
The formal charge on O is +1.
The formal charge on B is -1.
Learning Materials
Features
Discover
Chapter 2: Q.2-2-35_A (page 35)
Question: Calculate the formal charges on the atoms shown in red.
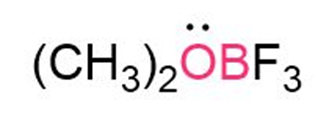
Answer
The formal charge on O is +1.
The formal charge on B is -1.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Which of the following pairs of structures represent resonance forms?
Question: Assign formal charges to the atoms in each of the following molecules:
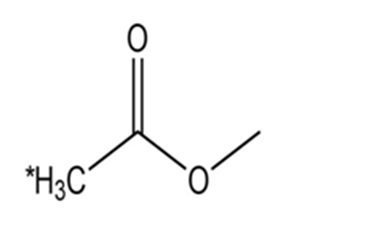
Draw the conjugate base for each compound below (the acidic hydrogen in each case is marked with an *).
(b)
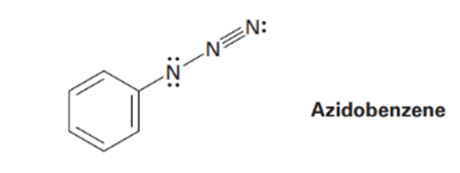
The azide functional group, which occurs in azido benzene, contains three adjacent nitrogen atoms. One resonance structure for azido benzene is shown. Draw three additional resonance structures, and assign appropriate formal charges to the atoms in all four.
Question: Draw as many resonance structures as you can for the following
species:
What do you think about this solution?
We value your feedback to improve our textbook solutions.