Chapter 2: Q.2-2-35_B (page 35)
Question: Calculate the formal charges on the atoms shown in red.
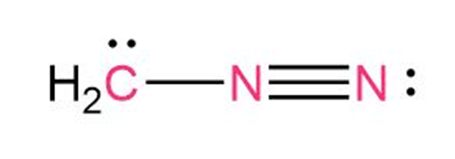
Short Answer
Answer
The formal charge on C is -1
The formal charge on N bonded with C is +1
The formal charge on N bonded with N is 0
Learning Materials
Features
Discover
Chapter 2: Q.2-2-35_B (page 35)
Question: Calculate the formal charges on the atoms shown in red.
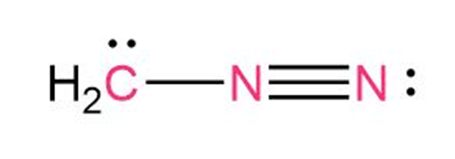
Answer
The formal charge on C is -1
The formal charge on N bonded with C is +1
The formal charge on N bonded with N is 0
All the tools & learning materials you need for study success - in one app.
Get started for free
Determine if each compound or ion below has a dipole moment.
(a)Carbonate ion
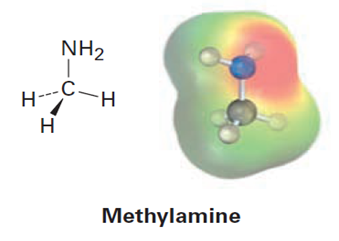
Look at the following electrostatic potential map of methylamine, a substance responsible for the odor of rotting fish, and tell the direction of polarization of the C-N bond:
Determine if each compound or ion below has a dipole moment.
(a)Carbonate ion
(b)
(c)
Question:
(A)The bond length is . What would the dipole moment of be if the molecule were ionic, ?
(B)The actual dipole moment of HCl is . What is the percent
ionic character of the bond?
:Which element in each of the following pairs is more electronegative?
(a)Li or H (b) B or Br (c) Cl or I (d) C or H
What do you think about this solution?
We value your feedback to improve our textbook solutions.