Chapter 2: Q. 15 P (page 47)
Ammonia, NH3, has=36, and acetone has=19. Will the following reaction take place to a significant extent?
Short Answer
The above reaction will take place to virtual completion due to large difference in a value.
Learning Materials
Features
Discover
Chapter 2: Q. 15 P (page 47)
Ammonia, NH3, has=36, and acetone has=19. Will the following reaction take place to a significant extent?
The above reaction will take place to virtual completion due to large difference in a value.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What is the Ka of HCN if its pKa=9.31?
Question: Assign formal charges to the atoms in each of the following molecules:
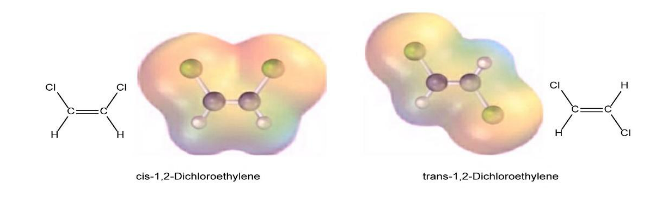
Question: Cis-1, 2-Dichloroethylene, and trans-1, 2-dichloroethylene are isomers, compounds with the same formula but different chemical structures. Look at the following electrostatic potential maps, and tell whether either compound has a dipole moment.
TheO-H hydrogen in acetic acid is more acidic than anyhydrogens. Explain this result using resonance structures.
Question: Phosgene, Cl2C=O , has a smaller dipole moment than formaldehyde, H2C=O , even though it contains electronegative chlorine atoms in place of hydrogen. Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.