Chapter 4: Q4-43E (page 114)
Draw the two chair conformations of trans-1-chloro-2-methylcyclohexane. Which is more stable?
Short Answer
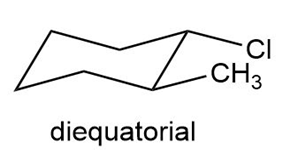
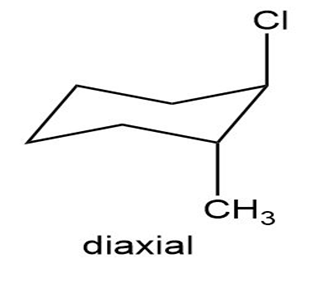
The diequitorial conformation of trans-1-chloro-2-methylcyclohexane is more stable.
Learning Materials
Features
Discover
Chapter 4: Q4-43E (page 114)
Draw the two chair conformations of trans-1-chloro-2-methylcyclohexane. Which is more stable?


The diequitorial conformation of trans-1-chloro-2-methylcyclohexane is more stable.
All the tools & learning materials you need for study success - in one app.
Get started for free
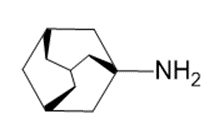
Question:Amantadine is an antiviral agent that is active against influenza type A infection. Draw a three-dimensional representation of amantadine, showing the chair cyclohexane rings.
Question:There are two different substances named trans-1,2-dimethylcyclopentane. What is the relationship between them?
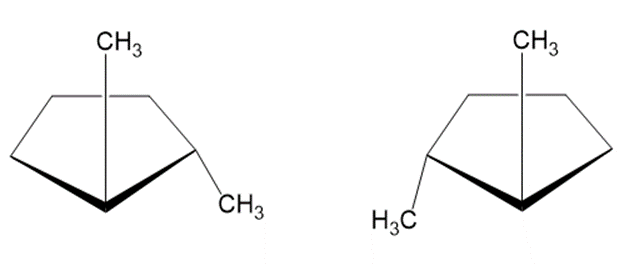
Draw three isomers of trans-1, 2-dichlorocyclobutane, and label them as either constitutional isomers or stereoisomers.
Cis-decalin is less stable than trans-decalin. Assume that the 1,3-diaxial interactions in cis-decalin are similar to those in axial methylcyclohexane [that is, one CH H interaction costs 3.8 kJ/mol (0.9 kcal/mol)], and calculate the magnitude of the energy difference between cis- and trans-decalin.
Draw two different chair conformations of trans-1,4 dimethylcyclohexane, and label all positions as axial or equatorial.
What do you think about this solution?
We value your feedback to improve our textbook solutions.