Chapter 4: Q4-31E (page 114)
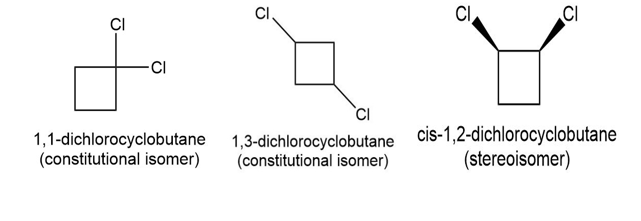
Draw three isomers of trans-1, 2-dichlorocyclobutane, and label them as either constitutional isomers or stereoisomers.
Short Answer
The three isomers oftrans-1, 2-dichlorocyclobutane are as follow;
Learning Materials
Features
Discover
Chapter 4: Q4-31E (page 114)
Draw three isomers of trans-1, 2-dichlorocyclobutane, and label them as either constitutional isomers or stereoisomers.
The three isomers oftrans-1, 2-dichlorocyclobutane are as follow;

All the tools & learning materials you need for study success - in one app.
Get started for free
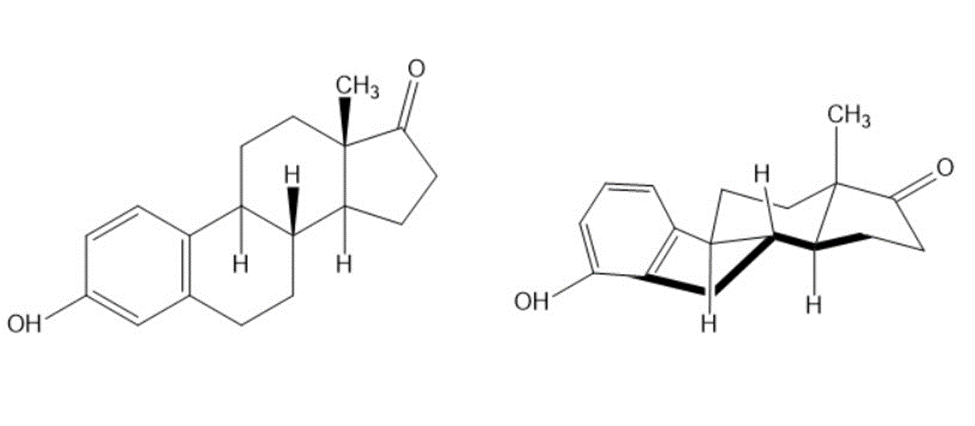
Look at the following structure of the female hormone estrone, and tell whether each of the two indicated ring-fusions is cis or trans.
A 1, 2-trans disubstituted cyclohexane must have either both groups axial or both groups equatorial. Explain.
Draw two different chair conformations of cyclohexanol (hydroxycyclohexane),showing all hydrogen atoms. Identify each position as axial orequatorial.
Why do you suppose an axial cyano (–CN) substituent causes practically no 1,3-diaxial steric strain (0.4 kJ/mol)? Use molecular models to help with your answer.
cis-1, 2-Dimethylcyclobutane is less stable than its trans isomer, but cis-1, 3-dimethylcyclobutane is more stable than its trans isomer. Draw the most stable conformations of both, and explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.