Chapter 4: Q.4-29E (page 114)
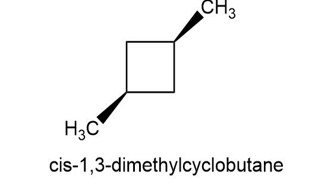
Draw a stereoisomer of trans-1, 3-dimethylcyclobutane.
Short Answer
There is only one stereoisomer of trans-1, 3-dimethylcyclobutane.
Learning Materials
Features
Discover
Chapter 4: Q.4-29E (page 114)
Draw a stereoisomer of trans-1, 3-dimethylcyclobutane.
There is only one stereoisomer of trans-1, 3-dimethylcyclobutane.

All the tools & learning materials you need for study success - in one app.
Get started for free
Which isomer is more stable, cis-decalin or trans-decalin? Explain.
Draw two different chair conformations of cyclohexanol (hydroxycyclohexane),showing all hydrogen atoms. Identify each position as axial orequatorial.
Two conformations of cis-1,3-dimethylcyclobutane are shown. What is the difference between them, and which do you think is likely to be more stable?

In light of your answer to Problem 4-48, draw the two chair conformations of 1,1,3-trimethylcyclohexane and estimate the amount of strain energy in each. Which conformation is favored?
Draw two different chair conformations of trans-1,4 dimethylcyclohexane, and label all positions as axial or equatorial.
What do you think about this solution?
We value your feedback to improve our textbook solutions.