Chapter 4: Q. 4-4-13P (page 104)
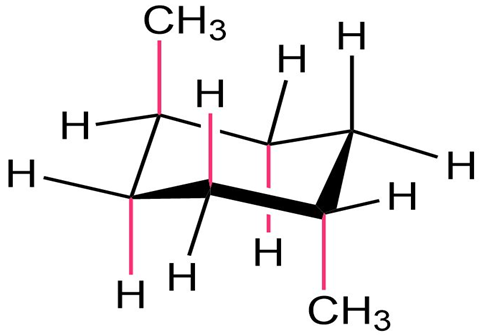
Draw two different chair conformations of trans-1,4 dimethylcyclohexane, and label all positions as axial or equatorial.
Short Answer
The two different chair conformations of trans-1,4 dimethylcyclohexane are:

Learning Materials
Features
Discover
Chapter 4: Q. 4-4-13P (page 104)
Draw two different chair conformations of trans-1,4 dimethylcyclohexane, and label all positions as axial or equatorial.
The two different chair conformations of trans-1,4 dimethylcyclohexane are:


All the tools & learning materials you need for study success - in one app.
Get started for free
How many eclipsing interactions would be present if cyclopentanewere planar? Assuming an energy cost of 4.0 kJ/mol for each eclipsing interaction,how much torsional strain would planar cyclopentane have? Since themeasured total strain of cyclopentane is 26 kJ/mol, how much of the torsionalstrain is relieved by puckering?
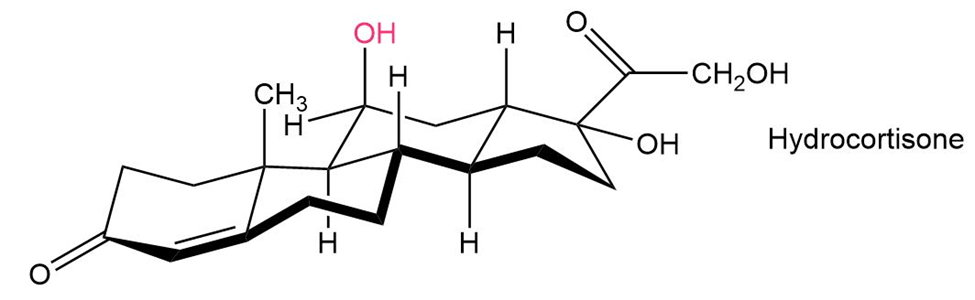
Hydrocortisone, a naturally occurring hormone produced in the adrenal glands, is often used to treat inflammation, severe allergies, and numerous other conditions. Is the indicated group axial or equatorial?
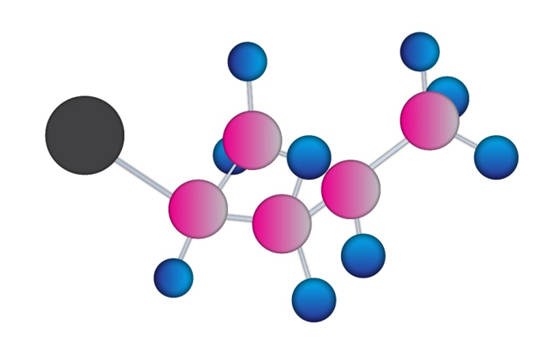
Name the following substances, including the cis- or trans- prefix (red-brown= Br):
Draw structures corresponding to the following IUPAC names:
a) 1,1 dimethyl cyclooctane
b) 3-cyclobutyl hexane
c) 1,2 -dichloro cyclopentane
d) 1,2- dibromo, 5- methylcyclohexane
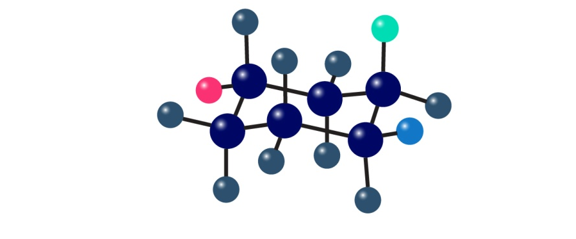
The following cyclohexane derivative has three red, green, and blue substituents. Identify each substituent as axial or equatorial, and identify each pair of relationships (red–blue, red-green, and blue-green) as cis or trans.
What do you think about this solution?
We value your feedback to improve our textbook solutions.