Chapter 4: Q4-4-2P (page 92)
Draw structures corresponding to the following IUPAC names:
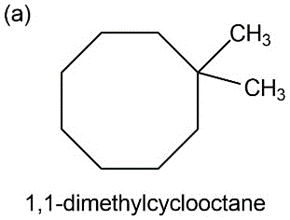
a) 1,1 dimethyl cyclooctane
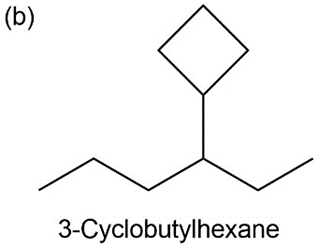
b) 3-cyclobutyl hexane
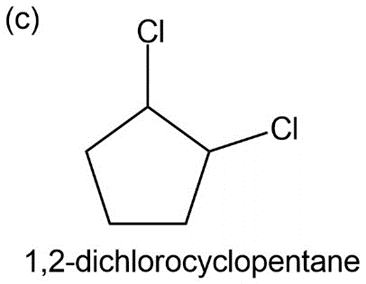
c) 1,2 -dichloro cyclopentane
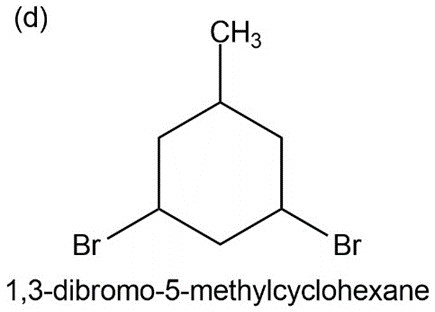
d) 1,2- dibromo, 5- methylcyclohexane
Short Answer
Learning Materials
Features
Discover
Chapter 4: Q4-4-2P (page 92)
Draw structures corresponding to the following IUPAC names:
a) 1,1 dimethyl cyclooctane
b) 3-cyclobutyl hexane
c) 1,2 -dichloro cyclopentane
d) 1,2- dibromo, 5- methylcyclohexane




All the tools & learning materials you need for study success - in one app.
Get started for free
A 1, 2-trans disubstituted cyclohexane must have either both groups axial or both groups equatorial. Explain.
Draw two different chair conformations of cyclohexanol (hydroxycyclohexane),showing all hydrogen atoms. Identify each position as axial orequatorial.
Draw the more stable chair conformation of the following molecules, and estimate the amount of strain in each: cis-1-tert-Butyl-4-ethylcyclohexane
In light of your answer to Problem 4-48, draw the two chair conformations of 1,1,3-trimethylcyclohexane and estimate the amount of strain energy in each. Which conformation is favored?
Name the following compound, identify each substituent as axial or equatorial, and tell whether the conformation shown is the more stable or less stable chair form
What do you think about this solution?
We value your feedback to improve our textbook solutions.