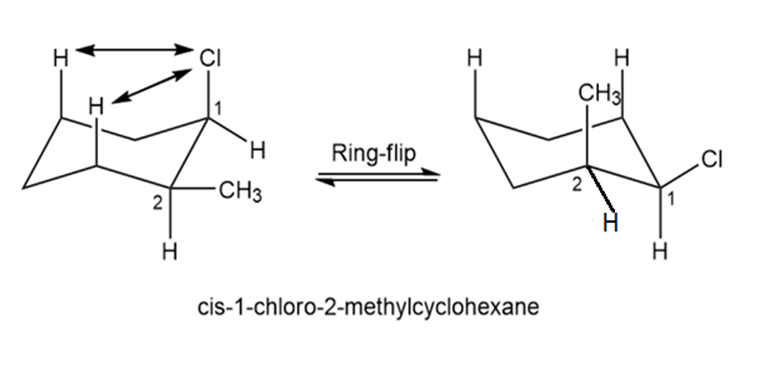
Chapter 4: Q4-42E (page 114)
Draw the two chair conformations of cis-1-chloro-2-methylcyclohexane. Which is more stable, and by how much?
Short Answer
The structure on the left-hand side is more stable than on the right-hand side.
Learning Materials
Features
Discover
Chapter 4: Q4-42E (page 114)
Draw the two chair conformations of cis-1-chloro-2-methylcyclohexane. Which is more stable, and by how much?
The structure on the left-hand side is more stable than on the right-hand side.

All the tools & learning materials you need for study success - in one app.
Get started for free
Each eclipsing interaction in ethane costs about 4.0 kJ/mol. Howmany such interactions are present in cyclopropane? What fraction of theoverall 115 kJ/mol (27.5 kcal/mol) strain energy of cyclopropane is due totorsional strain?
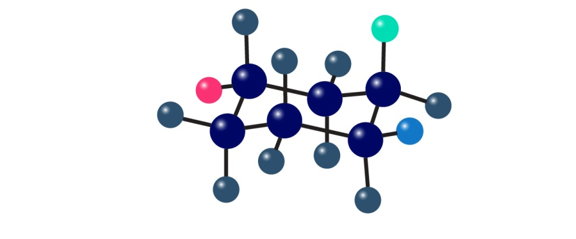
The following cyclohexane derivative has three red, green, and blue substituents. Identify each substituent as axial or equatorial, and identify each pair of relationships (red–blue, red-green, and blue-green) as cis or trans.
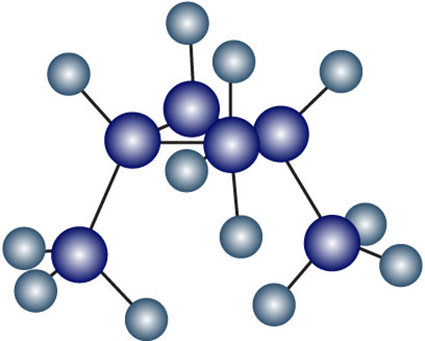
Two conformations of cis-1,3-dimethylcyclobutane are shown. What is the difference between them, and which do you think is likely to be more stable?

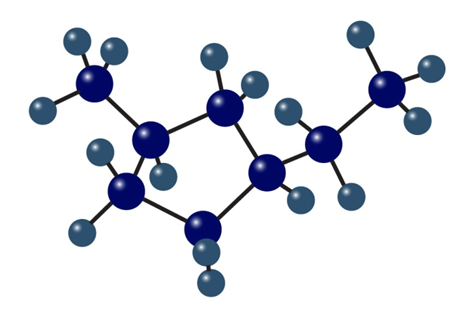
A 1, 2-cis disubstituted cyclohexane, such as cis-1, 2-dichlorocyclohexane, must have one group axial and one group equatorial. Explain.
Name the following cycloalkanes:
b. Name the following cycloalkanes:
What do you think about this solution?
We value your feedback to improve our textbook solutions.