The two chair conformations of 1,13-trimethyl cyclohexane is as follows:
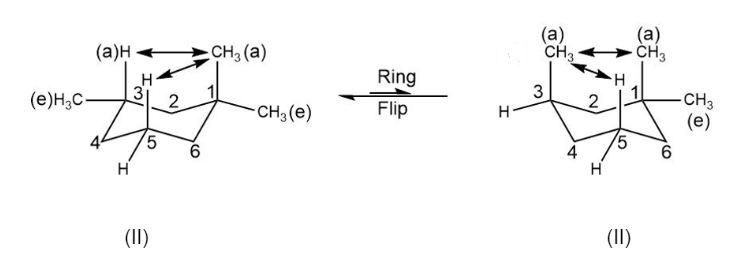
In structure II, there is one (CH3-CH3 ) 1,3-diaxial interaction thus it is higher in energy than structure I which has only (CH3-H ) 1,3-diaxial interaction. Thus, the structure I is more stable than structure II.
The structure I have diaxial interactions:
2(CH3-H) = 2 X 3.8 kJ/mol
= 7.6 kJ/mol
Structure II has greater diaxial interaction:
2(CH3-H) = 2 X 3.8 kJ/mol
= 7.6 kJ/mol
(CH3-CH3 ) = 5.4 kJ/mol
The total energy of the second conformer = (7.6+5.4) kJ/mol
The difference in the energy of two conformers = Energy of II-Energy of I
= 13.0-7.6 kJ/mol
= 5.4 kJ/mol
Thus, axial methyl groups produce 54 kJ/mol more steric strain in conformation II thus, conformation (I) is favored.




