Chapter 4: 4-20 (page 112)
Which isomer is more stable, cis-decalin or trans-decalin? Explain.
Short Answer
Trans-decalin is more stable as it shows fewer steric interactions.
Learning Materials
Features
Discover
Chapter 4: 4-20 (page 112)
Which isomer is more stable, cis-decalin or trans-decalin? Explain.
Trans-decalin is more stable as it shows fewer steric interactions.
All the tools & learning materials you need for study success - in one app.
Get started for free
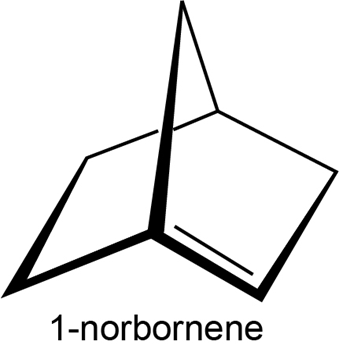
The German chemist J. Bredt proposed in 1935 that bicycloalkenes such as 1-norbornene, which have a double bond to the bridgehead carbon, are too strained to exist. Explain.
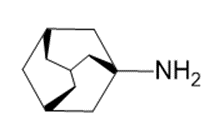
Question:Amantadine is an antiviral agent that is active against influenza type A infection. Draw a three-dimensional representation of amantadine, showing the chair cyclohexane rings.
Draw two constitutional isomers of.
Draw the five cycloalkanes with the formula .
Why is a 1, 3-cis disubstituted cyclohexane more stable than its trans isomer?
What do you think about this solution?
We value your feedback to improve our textbook solutions.