Chapter 4: Q60E (page 114)
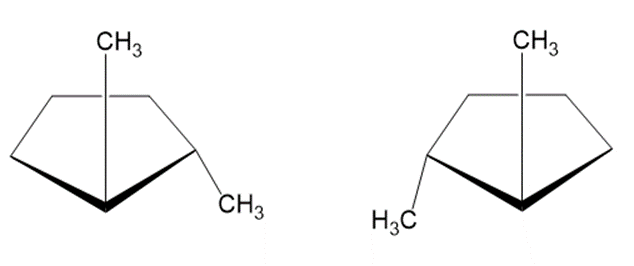
Question:There are two different substances named trans-1,2-dimethylcyclopentane. What is the relationship between them?
Short Answer
Answer
The two structures are enantiomers.
Learning Materials
Features
Discover
Chapter 4: Q60E (page 114)
Question:There are two different substances named trans-1,2-dimethylcyclopentane. What is the relationship between them?

Answer
The two structures are enantiomers.
All the tools & learning materials you need for study success - in one app.
Get started for free
Why do you suppose an axial cyano (–CN) substituent causes practically no 1,3-diaxial steric strain (0.4 kJ/mol)? Use molecular models to help with your answer.
Name the following substances, including the cis- or trans- prefix (redbrown= Br):
A 1, 2-trans disubstituted cyclohexane must have either both groups axial or both groups equatorial. Explain.
Why is a 1, 3-cis disubstituted cyclohexane more stable than its trans isomer?
Assume that you have a variety of cyclohexane substituted in the positions indicated. Identify the substituents as either axial or equatorial. For example, a 1, 2-cis relationship means that one substituent must be axial and one equatorial, whereas a 1, 2-trans relationship means that either substituent is axial or both are equatorial.
(a) 1, 3-Trans disubstituted (b) 1, 4-Cis disubstituted
(c) 1, 3-Cis disubstituted (d) 1, 5-Trans disubstituted
(e) 1, 5-Cis disubstituted (f) 1, 6-Trans disubstituted
What do you think about this solution?
We value your feedback to improve our textbook solutions.