Chapter 22: Q22-5P (page 734)
Show how you might prepare 1-penten-3-one from 3-pentanone.
Learning Materials
Features
Discover
Chapter 22: Q22-5P (page 734)
Show how you might prepare 1-penten-3-one from 3-pentanone.
All the tools & learning materials you need for study success - in one app.
Get started for free
How would you prepare the following ketones using an acetoacetic ester synthesis?

When an optically active carboxylic acid such as (R)-2-phenylpropanoic acid is brominated under Hell–Volhard–Zelinskii conditions, is the product optically active or racemic? Explain.
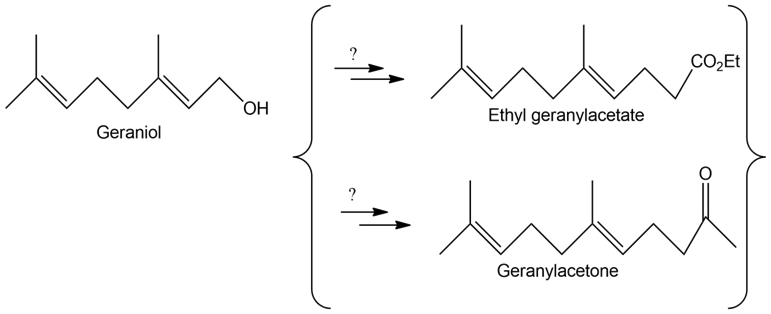
How might you convert geraniol into either ethyl geranylacetate or geranylacetone?
Would you expect optically active (S)-3-methylcyclohexanone to be racemized on acid or base treatment in the same way as 2-methylcyclohexanone (Problem 22-51)? Explain.
Draw a resonance structure of the acetonitrileanion,
andaccount for the acidity of nitriles.
What do you think about this solution?
We value your feedback to improve our textbook solutions.