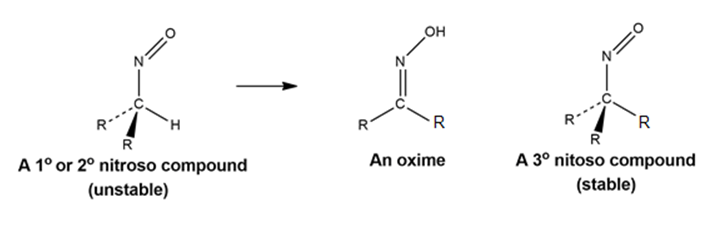
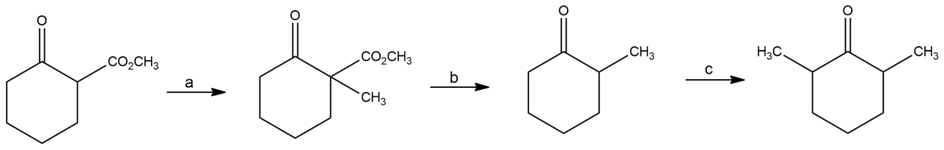
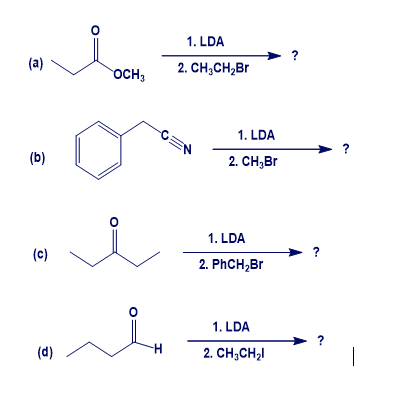
Chapter 22: Q22-62E (page 752)
Ketones react slowly with benzene selenenyl chloride in the presence of HCl to yield a-phenyl seleno ketones. Propose a mechanism for this acid-catalyzed a-substitution reaction.

Short Answer
The reaction mechanism starts with keto-enol tautomerism and ends with the loss of a proton.
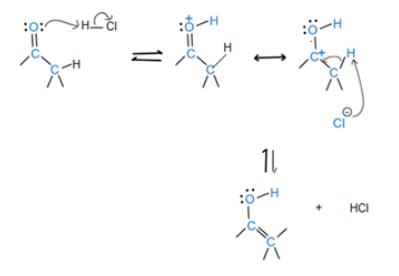
 The final product
The final product