Chapter 22: 45E (page 752)
How would you prepare the following ketones using an acetoacetic ester synthesis?

Short Answer
The given compounds are prepared by acetoacetic ester synthesis.
Learning Materials
Features
Discover
Chapter 22: 45E (page 752)
How would you prepare the following ketones using an acetoacetic ester synthesis?

The given compounds are prepared by acetoacetic ester synthesis.
All the tools & learning materials you need for study success - in one app.
Get started for free
Amino acids can also be prepared by a two-step sequence that involves Hell–Volhard–Zelinskii reaction of a carboxylic acid followed by treatment with ammonia. Show how you would prepare leucine, (CH3)2CHCH2CH(NH2)CO2H, and identify the mechanism of the second step.
The two isomers cis- and trans-4-tert-butyl-2-methylcyclohexanone are interconverted by base treatment. Which isomer do you think is more stable, and why?
Which, if any, of the following compounds can be prepared by an acetoacetic ester synthesis? Explain.
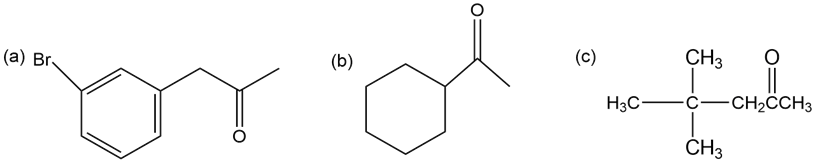
Heating carvone with aqueous sulfuric acid converts it into carvacrol. Propose a mechanism for the isomerization.
Draw a resonance structure of the acetonitrileanion,
andaccount for the acidity of nitriles.
What do you think about this solution?
We value your feedback to improve our textbook solutions.