Chapter 22: Q36E (page 752)
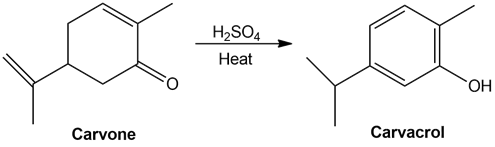
Heating carvone with aqueous sulfuric acid converts it into carvacrol. Propose a mechanism for the isomerization.
Short Answer
Learning Materials
Features
Discover
Chapter 22: Q36E (page 752)
Heating carvone with aqueous sulfuric acid converts it into carvacrol. Propose a mechanism for the isomerization.


All the tools & learning materials you need for study success - in one app.
Get started for free
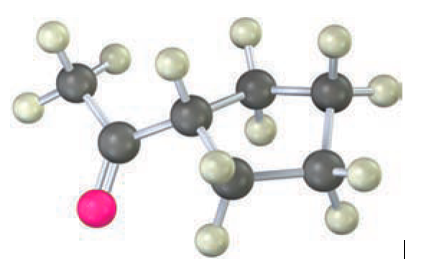
Question: How would you prepare the following compound using an acetoacetic ester synthesis?
Which, if any, of the following compounds can be prepared by a malonic ester synthesis? Show the alkyl halide you would use in each case.
(a)Ethyl pentanoate (b)Ethyl 3-methylbutanoate
(c)Ethyl 2-methylbutanoate (d) Ethyl 2,2-dimethylpropanoate
Would you expect optically active (S)-3-methylcyclohexanone to be racemized on acid or base treatment in the same way as 2-methylcyclohexanone (Problem 22-51)? Explain.
Write the complete mechanism for the deuteration of acetone on treatment with D3O+.
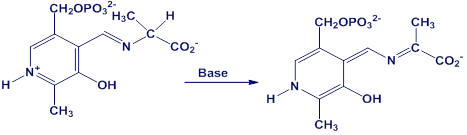
Using curved arrows, propose a mechanism for the following reaction, one of the steps in the metabolism of the amino acid alanine.
What do you think about this solution?
We value your feedback to improve our textbook solutions.