Chapter 22: 15P (page 728)
Question: How would you prepare the following compound using an acetoacetic ester synthesis?
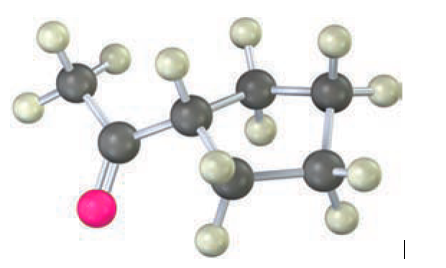
Short Answer
Learning Materials
Features
Discover
Chapter 22: 15P (page 728)
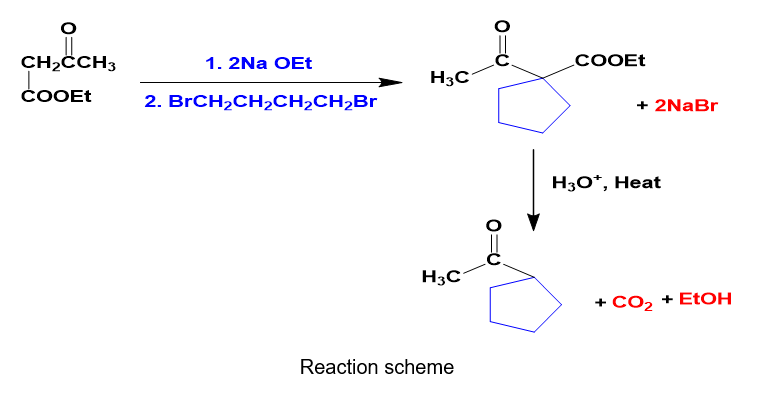
Question: How would you prepare the following compound using an acetoacetic ester synthesis?


All the tools & learning materials you need for study success - in one app.
Get started for free
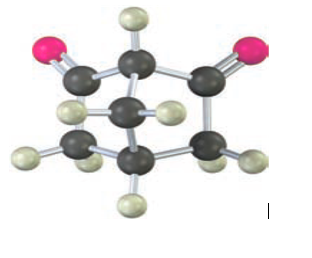
Unlike most β-diketones, the following β-diketone has no detectable enol content and is about as acidic as acetone. Explain.
Rank the following compounds in order of increasing acidity:
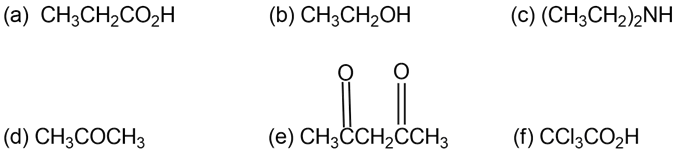
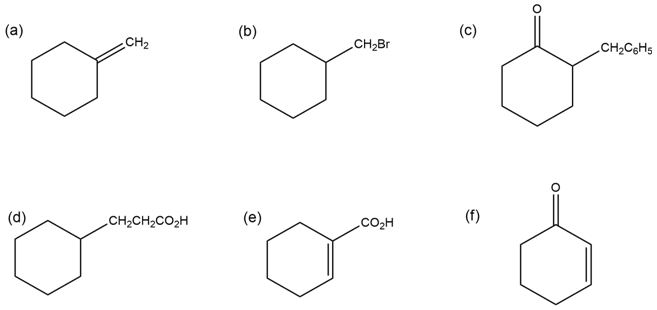
How would you synthesize the following compounds from cyclohexanone?
More than one step may be required.
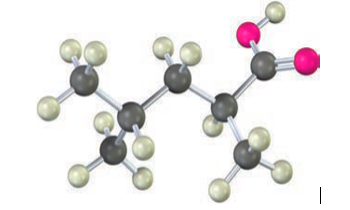
How could you use a malonic ester synthesis to prepare the following compound?
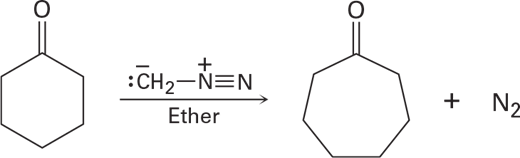
Treatment of a cyclic ketone with diazomethane is a method for accomplishing a ring-expansion reaction.For example, treatment of cyclohexanone with diazomethane yields cycloheptanone. Propose a mechanism.
What do you think about this solution?
We value your feedback to improve our textbook solutions.