Chapter 22: 58E (page 752)
How would you synthesize the following compounds from cyclohexanone?
More than one step may be required.
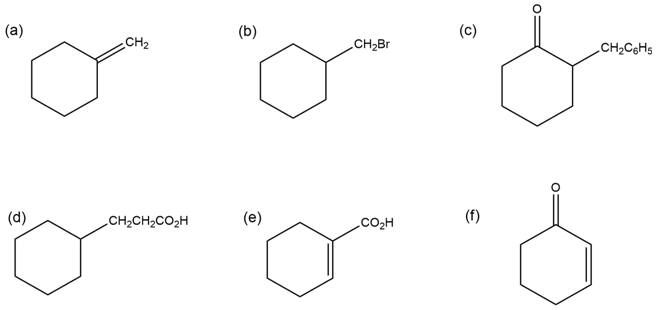
Short Answer
a)
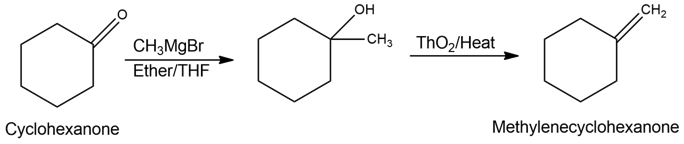
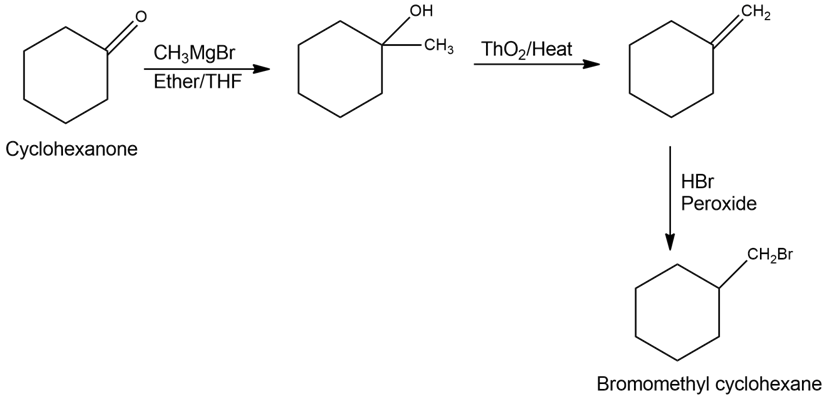
b)
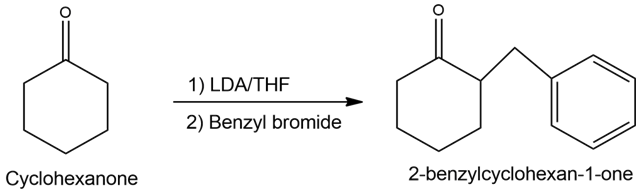
c)
d)
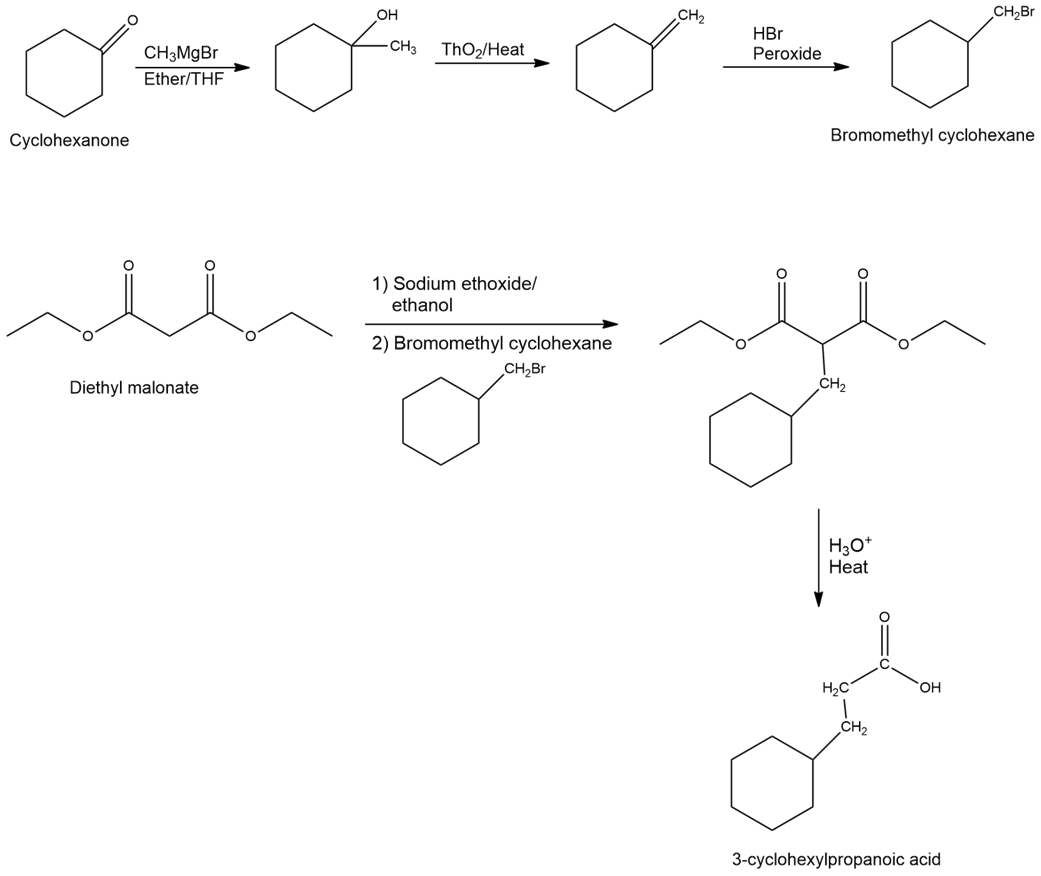
e)
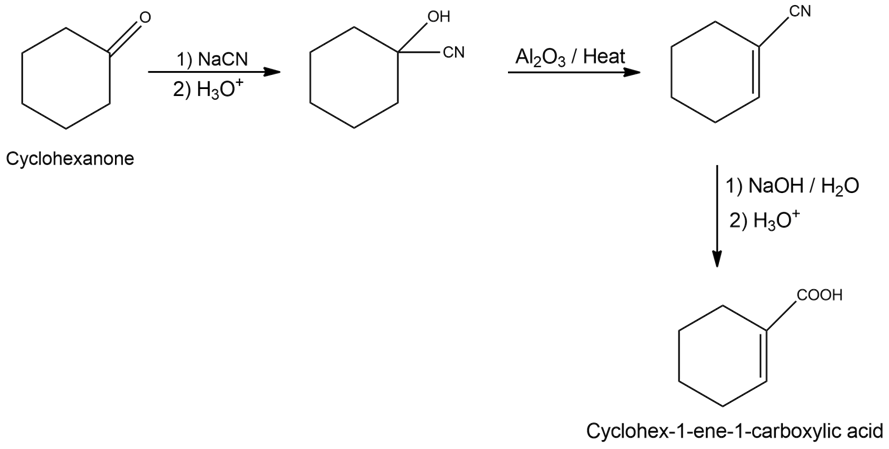
f)
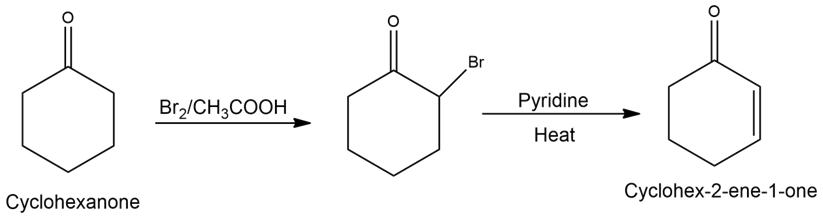
Learning Materials
Features
Discover
Chapter 22: 58E (page 752)
How would you synthesize the following compounds from cyclohexanone?
More than one step may be required.

a)

b)

c)

d)

e)

f)

All the tools & learning materials you need for study success - in one app.
Get started for free
When an optically active carboxylic acid such as (R)-2-phenylpropanoic acid is brominated under Hell–Volhard–Zelinskii conditions, is the product optically active or racemic? Explain.
Predict the product(s) and provide the mechanism for each reaction below.
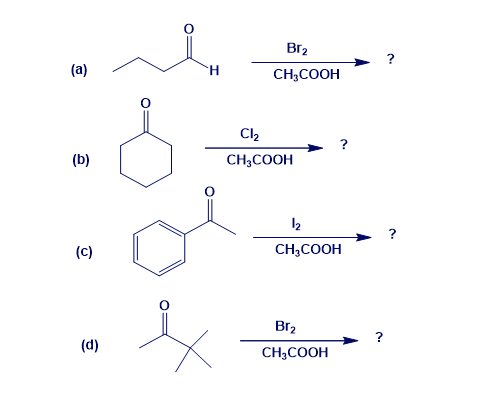
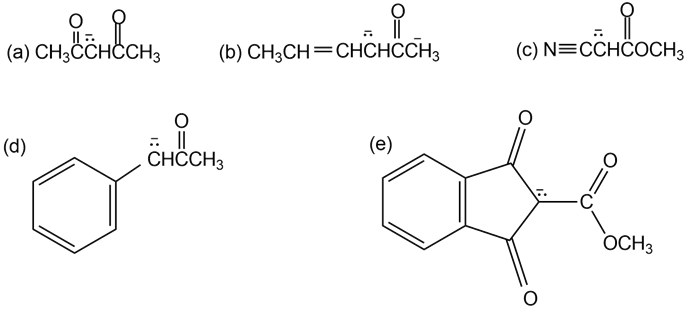
Write resonance structures for the following anions:
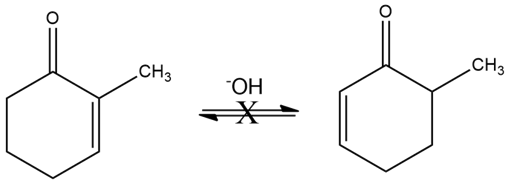
Although 2-substituted 2-cyclopentenones are in a base-catalyzed equilibrium with their 5-substituted 2-cyclopentenone isomers (Problem 22-55), the analogous isomerization is not observed for 2-substituted 2-cyclohexenones. Explain.
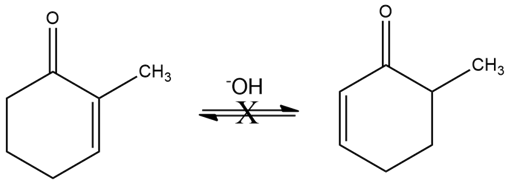
Although 2-substituted 2-cyclopentenones are in a base-catalyzed equilibrium with their 5-substituted 2-cyclopentenone isomers (Problem 22-55), the analogous isomerization is not observed for 2-substituted 2-cyclohexenones. Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.