Chapter 22: Q39E (page 752)
Write resonance structures for the following anions:
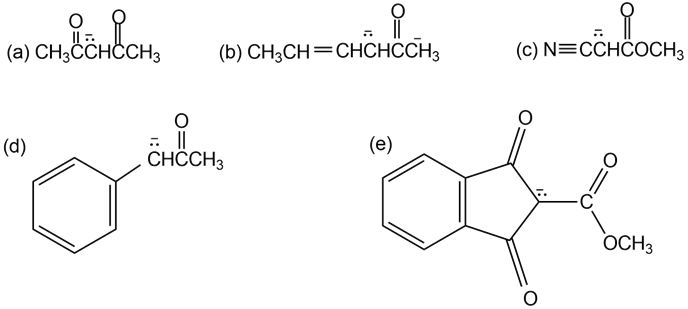
Short Answer
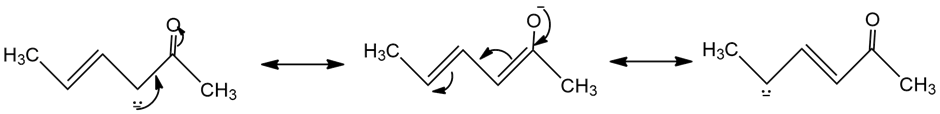
 Resonance forms of (a)
Resonance forms of (a)
Resonance forms of (b)
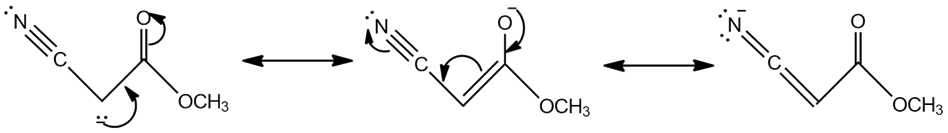
Resonance forms of (c)
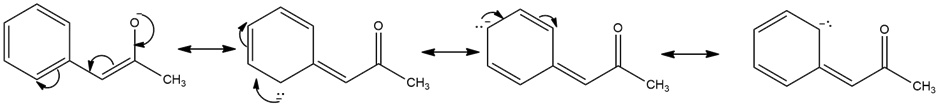
Resonance forms of (d)
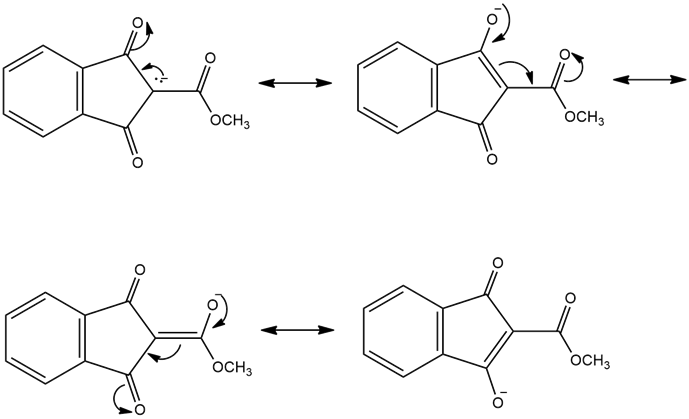
Resonance forms of (e)
Learning Materials
Features
Discover
Chapter 22: Q39E (page 752)
Write resonance structures for the following anions:

 Resonance forms of (a)
Resonance forms of (a)

Resonance forms of (b)

Resonance forms of (c)

Resonance forms of (d)

Resonance forms of (e)
All the tools & learning materials you need for study success - in one app.
Get started for free
Unlike most β-diketones, the following β-diketone has no detectable enol content and is about as acidic as acetone. Explain.
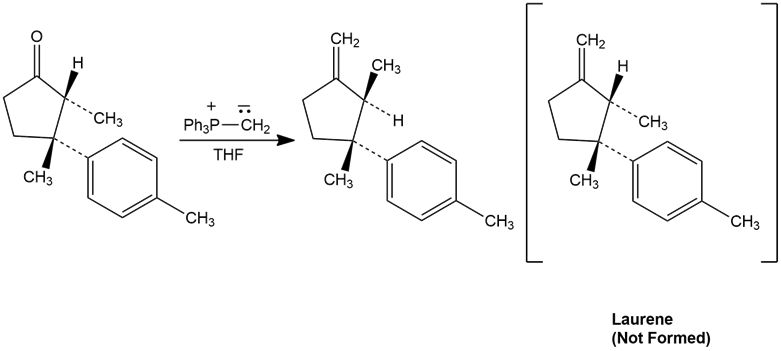
The final step in attempting to synthesize Laurene, a hydrocarbon isolated from the marine alga Laurencia glandulifera,involved the Wittig reaction shown. The product obtained, however, was not Laurene but an isomer. Propose a mechanism to account for these unexpected results.
How would you prepare the following ketones using an acetoacetic ester synthesis?

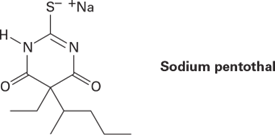
Sodium pentothal is a short-acting barbiturate derivative used as a general anesthetic and known as a truth serum in popular culture. It is synthesized like other barbiturates (see the Something Extra at the end of this chapter), using thiourea, (H2N)2C=S, in place of urea. How would you synthesize sodium pentothal?
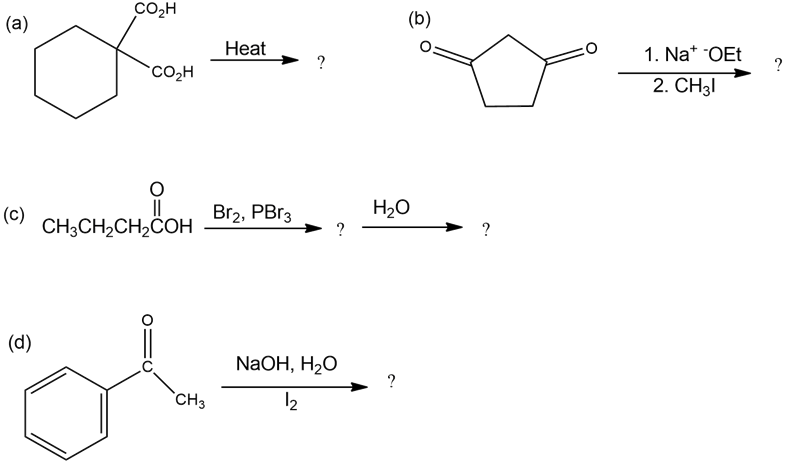
Predict the product(s) of the following reactions:
What do you think about this solution?
We value your feedback to improve our textbook solutions.