Chapter 22: Q42E (page 752)
Predict the product(s) of the following reactions:
Short Answer

Chemical reaction (a)
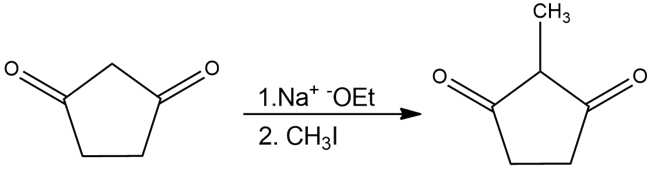
Chemical reaction (b)
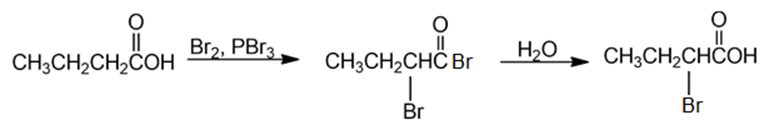
Chemical reaction (c)
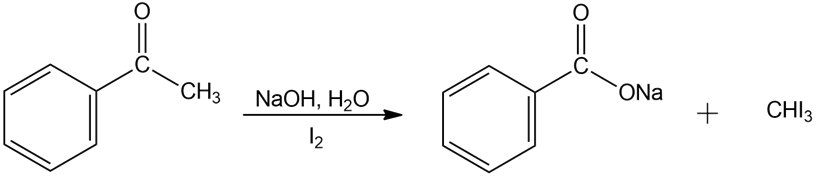
Chemical reaction (d)
Learning Materials
Features
Discover
Chapter 22: Q42E (page 752)
Predict the product(s) of the following reactions:


Chemical reaction (a)
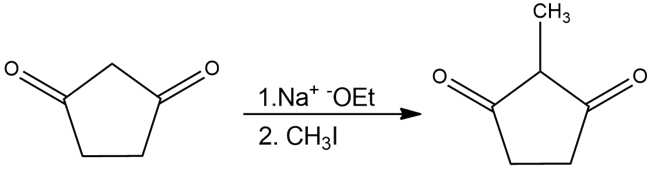
Chemical reaction (b)

Chemical reaction (c)

Chemical reaction (d)
All the tools & learning materials you need for study success - in one app.
Get started for free
How would you prepare the following ketones using an acetoacetic ester synthesis?

Show the steps in preparing each of the following substances using either a malonic ester synthesis or an acetoacetic ester synthesis:
.
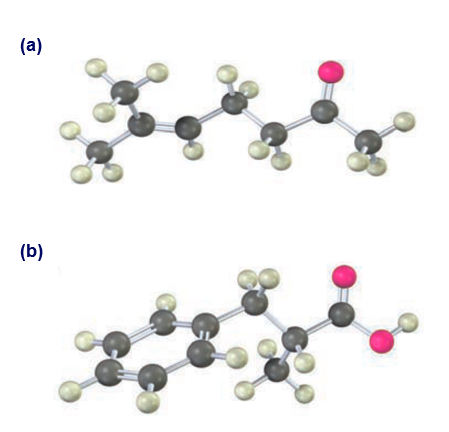
The two isomers cis- and trans-4-tert-butyl-2-methylcyclohexanone are interconverted by base treatment. Which isomer do you think is more stable, and why?
Question: Identify the most acidic Hydrogens in each of the following molecules:
(a)CH3CH2CHO
(b)(CH3)3CCOCH3
(c)CH3CO2H
(d)Benzamide
(e)CH3CH2CH2CN
(f)CH3CON(CH3)2
Draw a resonance structure of the acetonitrileanion,
andaccount for the acidity of nitriles.
What do you think about this solution?
We value your feedback to improve our textbook solutions.