Chapter 22: Q3P (page 728)
Draw structures for all monoenol forms of the following molecule. Which would you expect to be the most stable? Explain.
Learning Materials
Features
Discover
Chapter 22: Q3P (page 728)
Draw structures for all monoenol forms of the following molecule. Which would you expect to be the most stable? Explain.
All the tools & learning materials you need for study success - in one app.
Get started for free
When an optically active carboxylic acid such as (R)-2-phenylpropanoic acid is brominated under Hell–Volhard–Zelinskii conditions, is the product optically active or racemic? Explain.
Which, if any, of the following compounds can be prepared by a malonic ester synthesis? Show the alkyl halide you would use in each case.
(a)Ethyl pentanoate (b)Ethyl 3-methylbutanoate
(c)Ethyl 2-methylbutanoate (d) Ethyl 2,2-dimethylpropanoate
Amino acids can also be prepared by a two-step sequence that involves Hell–Volhard–Zelinskii reaction of a carboxylic acid followed by treatment with ammonia. Show how you would prepare leucine, (CH3)2CHCH2CH(NH2)CO2H, and identify the mechanism of the second step.
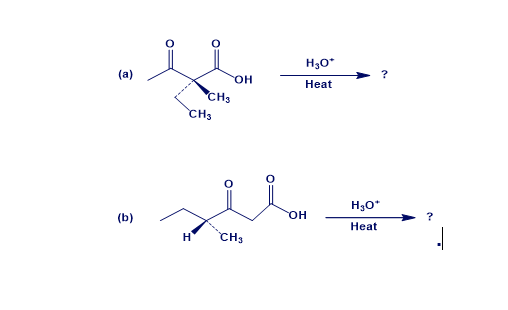
The two optically beta-keto acids below were decarboxylated using the conditions typically used for the acetoacetate synthesis. Will the ketone products also be optically active? Provide the complete mechanism to explain your answer.
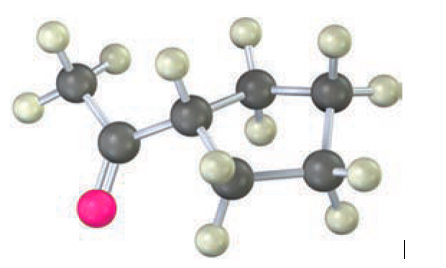
Question: How would you prepare the following compound using an acetoacetic ester synthesis?
What do you think about this solution?
We value your feedback to improve our textbook solutions.