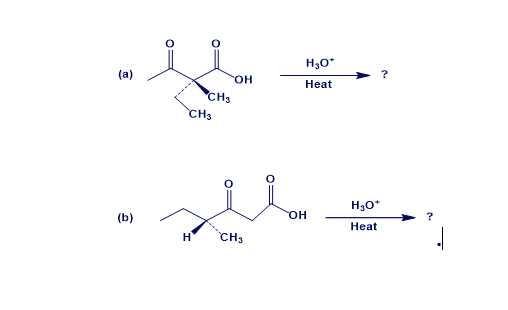
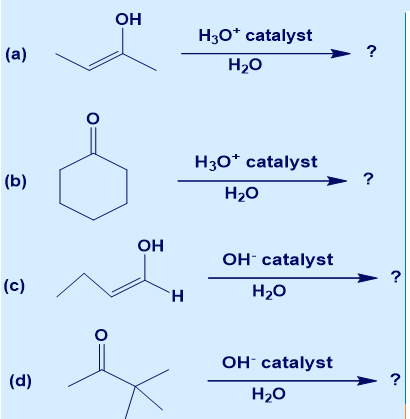
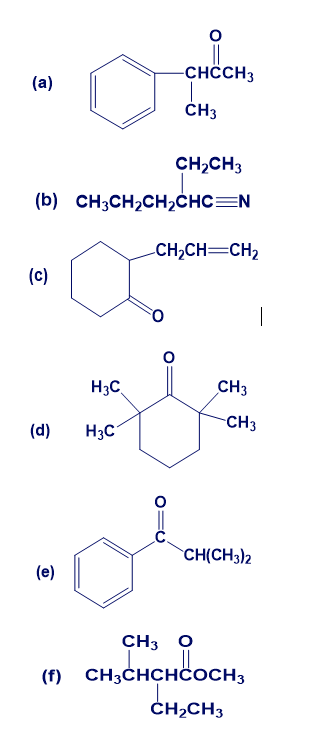
Chapter 22: 53E (page 752)
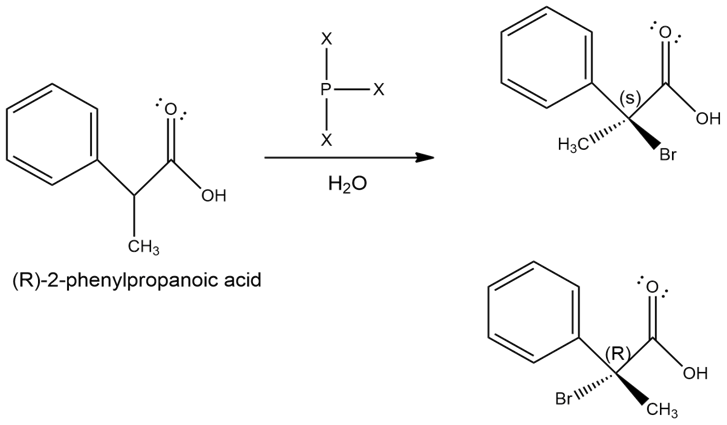
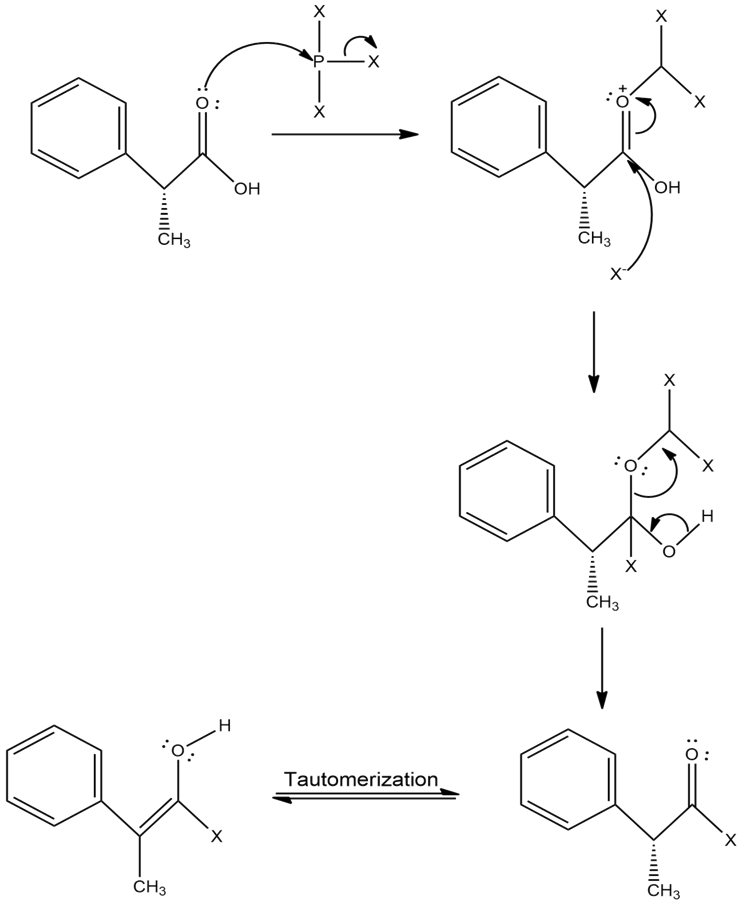
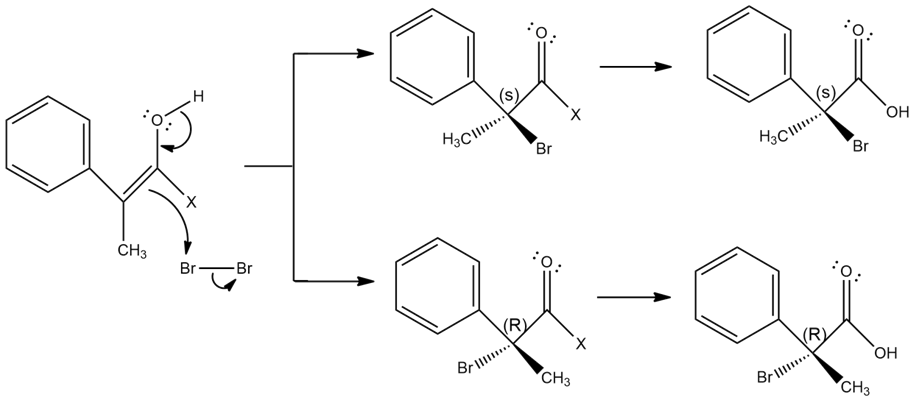
When an optically active carboxylic acid such as (R)-2-phenylpropanoic acid is brominated under Hell–Volhard–Zelinskii conditions, is the product optically active or racemic? Explain.
Short Answer
The compound, (R)-2-phenylpropanoic acid is brominated under Hell–Volhard–Zelinskii conditions produces racemic mixture.