Chapter 22: Q8P (page 738)
Draw a resonance structure of the acetonitrileanion,
andaccount for the acidity of nitriles.
Short Answer
The acidity of nitriles occurs due to the removal of a proton and its stable conjugate base stabilised by resonance.
Learning Materials
Features
Discover
Chapter 22: Q8P (page 738)
Draw a resonance structure of the acetonitrileanion,
andaccount for the acidity of nitriles.
The acidity of nitriles occurs due to the removal of a proton and its stable conjugate base stabilised by resonance.
All the tools & learning materials you need for study success - in one app.
Get started for free
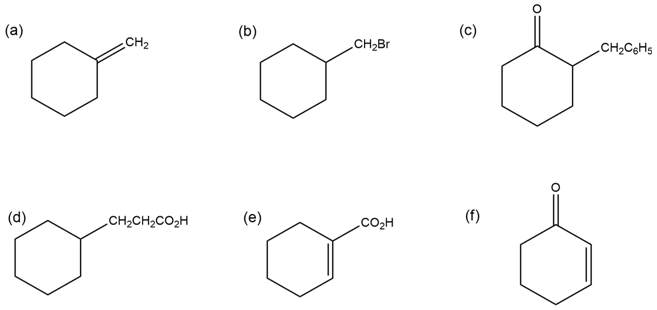
How would you synthesize the following compounds from cyclohexanone?
More than one step may be required.
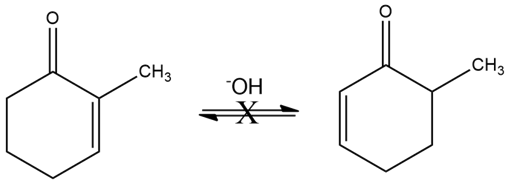
Although 2-substituted 2-cyclopentenones are in a base-catalyzed equilibrium with their 5-substituted 2-cyclopentenone isomers (Problem 22-55), the analogous isomerization is not observed for 2-substituted 2-cyclohexenones. Explain.
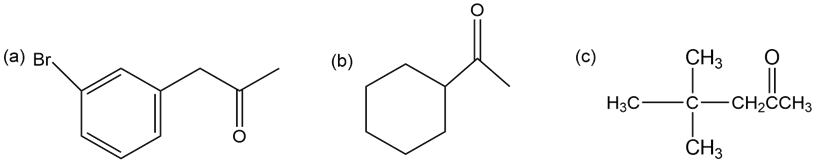
Which, if any, of the following compounds can be prepared by an acetoacetic ester synthesis? Explain.
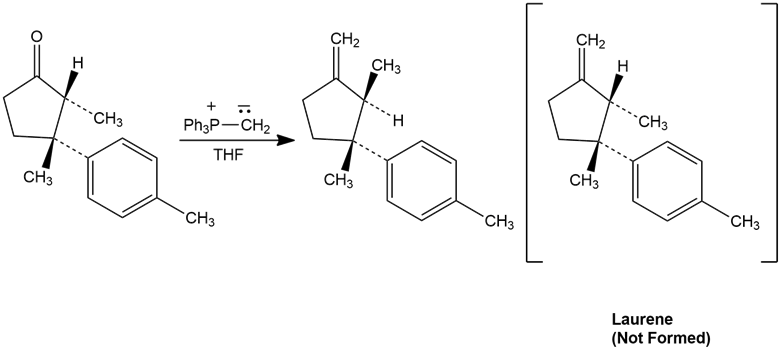
The final step in attempting to synthesize Laurene, a hydrocarbon isolated from the marine alga Laurencia glandulifera,involved the Wittig reaction shown. The product obtained, however, was not Laurene but an isomer. Propose a mechanism to account for these unexpected results.
Question: Identify the most acidic Hydrogens in each of the following molecules:
(a)CH3CH2CHO
(b)(CH3)3CCOCH3
(c)CH3CO2H
(d)Benzamide
(e)CH3CH2CH2CN
(f)CH3CON(CH3)2
What do you think about this solution?
We value your feedback to improve our textbook solutions.