Chapter 22: 12P (page 728)
How could you use a malonic ester synthesis to prepare the following compound?
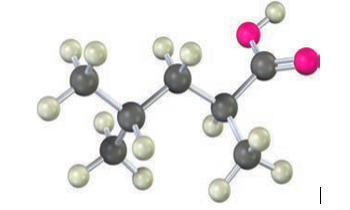
Short Answer
Learning Materials
Features
Discover
Chapter 22: 12P (page 728)
How could you use a malonic ester synthesis to prepare the following compound?


All the tools & learning materials you need for study success - in one app.
Get started for free
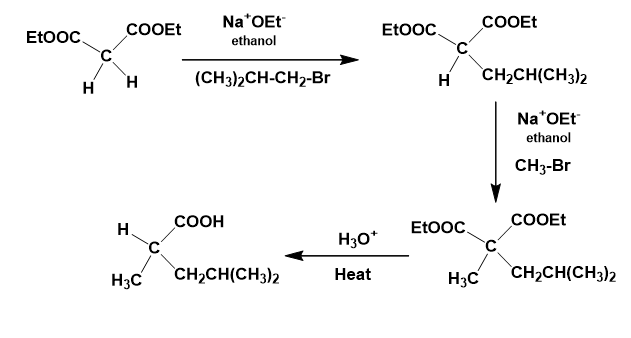
How would you prepare the following compounds using either an Acetoacetic ester synthesis or a malonic ester synthesis?

Would you expect optically active (S)-3-methylcyclohexanone to be racemized on acid or base treatment in the same way as 2-methylcyclohexanone (Problem 22-51)? Explain.
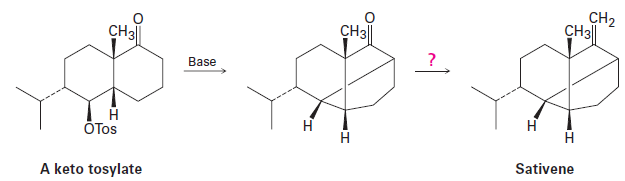
The key step in a reported laboratory synthesis of sativene, a hydrocarbon isolated from the mold Helminthosporium sativum, involves the following base treatment of a keto tosylate.
What kind of reaction is occurring? How would you complete the synthesis?
Question: Identify the most acidic Hydrogens in each of the following molecules:
(a)CH3CH2CHO
(b)(CH3)3CCOCH3
(c)CH3CO2H
(d)Benzamide
(e)CH3CH2CH2CN
(f)CH3CON(CH3)2
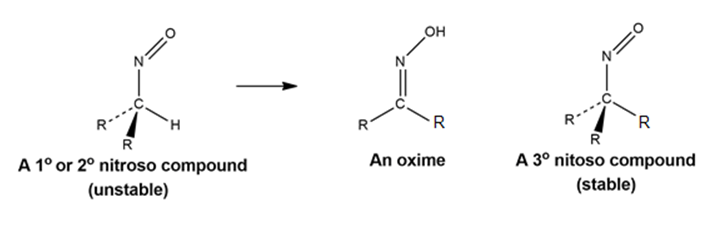
All attempts to isolate primary and secondary nitroso compounds result solely in the formation of oximes. Tertiary nitroso compounds, however, are stable. Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.