Chapter 22: Q32E (page 752)
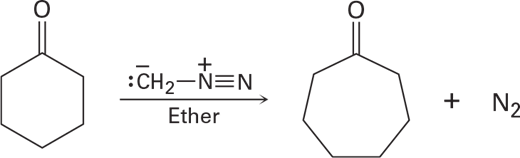
Treatment of a cyclic ketone with diazomethane is a method for accomplishing a ring-expansion reaction.For example, treatment of cyclohexanone with diazomethane yields cycloheptanone. Propose a mechanism.
Short Answer
Learning Materials
Features
Discover
Chapter 22: Q32E (page 752)
Treatment of a cyclic ketone with diazomethane is a method for accomplishing a ring-expansion reaction.For example, treatment of cyclohexanone with diazomethane yields cycloheptanone. Propose a mechanism.


All the tools & learning materials you need for study success - in one app.
Get started for free
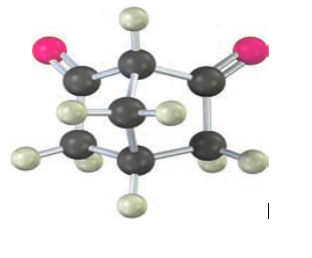
Unlike most β-diketones, the following β-diketone has no detectable enol content and is about as acidic as acetone. Explain.
Draw a resonance structure of the acetonitrileanion,
andaccount for the acidity of nitriles.
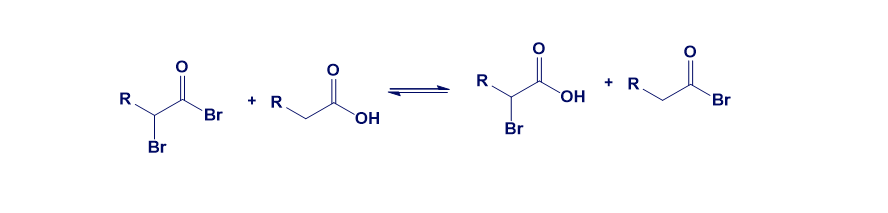
In the Hell–Volhard–Zelinskii reaction, only a catalytic amount of PBr3 is necessary because of the equilibrium below. Review the mechanism for the reaction of a carboxylic acid with thionyl chloride and propose a mechanism for the equilibrium.
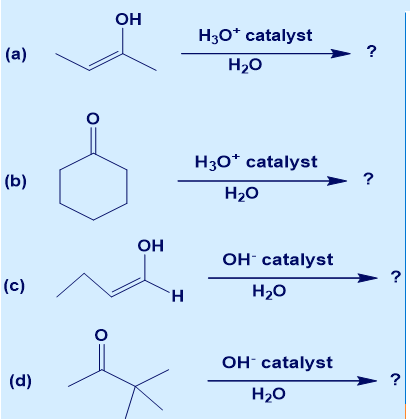
For each reaction below, give the corresponding keto/enol tautomer and provide the complete mechanism.
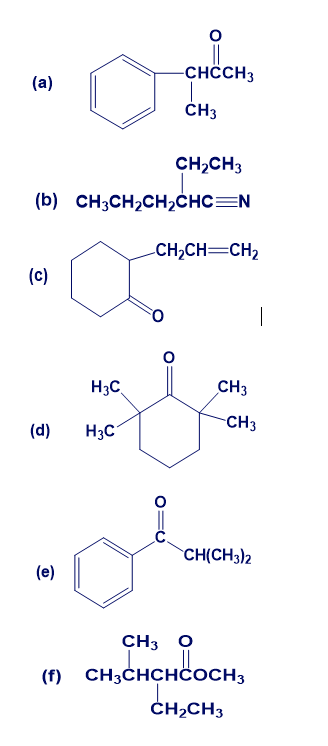
Show how you might prepare the following compounds using an alkylation reaction as the key step:
What do you think about this solution?
We value your feedback to improve our textbook solutions.