Chapter 22: Q28E (page 752)
Using curved arrows, propose a mechanism for the following reaction, one of the steps in the metabolism of the amino acid alanine.
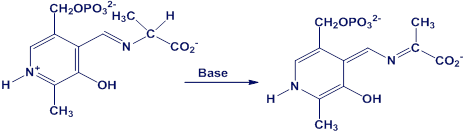
Short Answer
You need to propose a mechanism for the given reaction.
Learning Materials
Features
Discover
Chapter 22: Q28E (page 752)
Using curved arrows, propose a mechanism for the following reaction, one of the steps in the metabolism of the amino acid alanine.

You need to propose a mechanism for the given reaction.
All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you might prepare 1-penten-3-one from 3-pentanone.
Draw structures for all monoenol forms of the following molecule. Which would you expect to be the most stable? Explain.
The two isomers cis- and trans-4-tert-butyl-2-methylcyclohexanone are interconverted by base treatment. Which isomer do you think is more stable, and why?
If methanol rather than water is added at the end of a Hell–Volhard–Zelinskii reaction, an ester rather than an acid is produced. Show how you would carry out the following transformation, and propose a mechanism for the ester forming step.
Although 2-substituted 2-cyclopentenones are in a base-catalyzed equilibrium with their 5-substituted 2-cyclopentenone isomers (Problem 22-55), the analogous isomerization is not observed for 2-substituted 2-cyclohexenones. Explain.
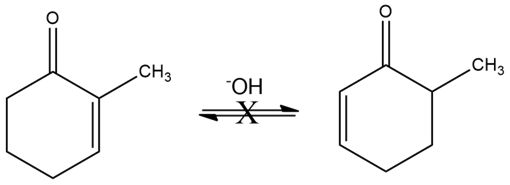
What do you think about this solution?
We value your feedback to improve our textbook solutions.