Chapter 22: Q4P (page 734)
Write the complete mechanism for the deuteration of acetone on treatment with D3O+.
Learning Materials
Features
Discover
Chapter 22: Q4P (page 734)
Write the complete mechanism for the deuteration of acetone on treatment with D3O+.
All the tools & learning materials you need for study success - in one app.
Get started for free
How would you synthesize the following compounds from cyclohexanone?
More than one step may be required.
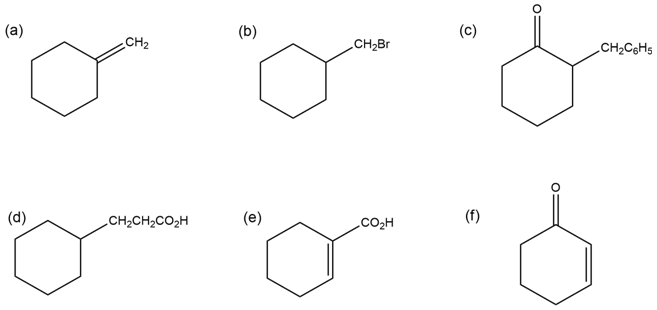
How many acidic Hydrogens does each of the molecules listed in Problem 22-1 have? Identify them.
Show how you might prepare 1-penten-3-one from 3-pentanone.
If methanol rather than water is added at the end of a Hell–Volhard–Zelinskii reaction, an ester rather than an acid is produced. Show how you would carry out the following transformation, and propose a mechanism for the ester forming step.
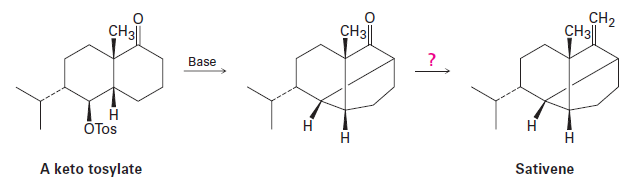
The key step in a reported laboratory synthesis of sativene, a hydrocarbon isolated from the mold Helminthosporium sativum, involves the following base treatment of a keto tosylate.
What kind of reaction is occurring? How would you complete the synthesis?
What do you think about this solution?
We value your feedback to improve our textbook solutions.