Chapter 22: 48E (page 752)
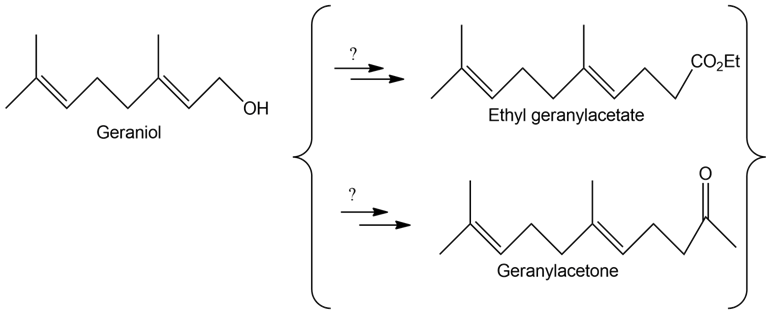
How might you convert geraniol into either ethyl geranylacetate or geranylacetone?
Short Answer
The geraniol is converted into geranylacetate and geranylacetone.
Learning Materials
Features
Discover
Chapter 22: 48E (page 752)
How might you convert geraniol into either ethyl geranylacetate or geranylacetone?

The geraniol is converted into geranylacetate and geranylacetone.
All the tools & learning materials you need for study success - in one app.
Get started for free
One way to determine the number of acidic hydrogens in a molecule is to treat the compound with NaOD in D2O, isolate the product, and determine its molecular weight by mass spectrometry. For example, if cyclohexanone is treated with NaOD in D2O, the product has MW =102. Explain how this method works.
How many acidic Hydrogens does each of the molecules listed in Problem 22-1 have? Identify them.
Amino acids can also be prepared by a two-step sequence that involves Hell–Volhard–Zelinskii reaction of a carboxylic acid followed by treatment with ammonia. Show how you would prepare leucine, (CH3)2CHCH2CH(NH2)CO2H, and identify the mechanism of the second step.
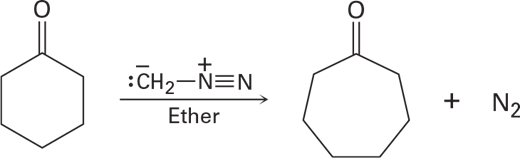
Treatment of a cyclic ketone with diazomethane is a method for accomplishing a ring-expansion reaction.For example, treatment of cyclohexanone with diazomethane yields cycloheptanone. Propose a mechanism.
Ketones react slowly with benzene selenenyl chloride in the presence of HCl to yield a-phenyl seleno ketones. Propose a mechanism for this acid-catalyzed a-substitution reaction.

What do you think about this solution?
We value your feedback to improve our textbook solutions.