Chapter 15: Q15-15-26E (page 477)
Phenanthrene has five resonance structures, one of which is shown. Draw the other four.
Short Answer
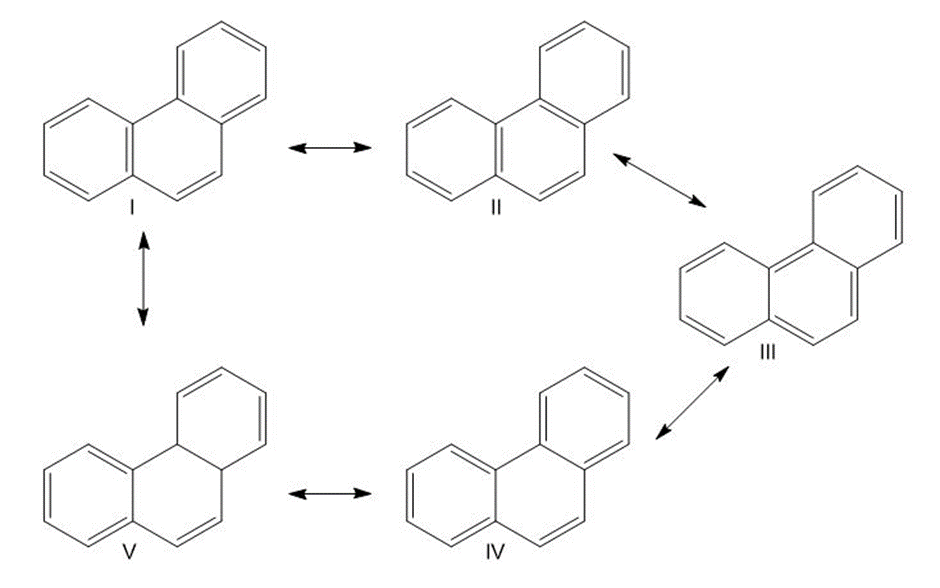
Five possible resonance structures of phenanthrene
Learning Materials
Features
Discover
Chapter 15: Q15-15-26E (page 477)
Phenanthrene has five resonance structures, one of which is shown. Draw the other four.

Five possible resonance structures of phenanthrene
All the tools & learning materials you need for study success - in one app.
Get started for free
Give IUPAC names for the following compounds
a)
Ribavirin, an antiviral agent used against hepatitis C and viral pneumonia, contains a 1,2,4-triazole ring. Why is the ring aromatic?
 Ribavirin
Ribavirin
Calicene, like azulene (Problem 15-17), has an unusually large dipole moment for a hydrocarbon. Explain, using resonance structures.
 Calicene
Calicene
After the reaction below, the chemical shift ofmoves downfield from 6.98 ppm to 7.30 ppm. Explain.
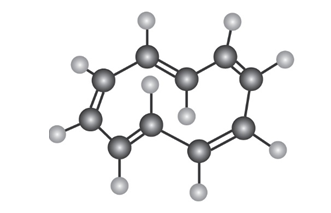
To be aromatic, a molecule must have 4n+2 p electrons and must have a planar, monocyclic system of conjugation. Cyclodecapentaene fulfills one of these criteria but not the other and has resisted all attempts at synthesis. Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.