Chapter 15: Q15-38E (page 477)
Ribavirin, an antiviral agent used against hepatitis C and viral pneumonia, contains a 1,2,4-triazole ring. Why is the ring aromatic?
 Ribavirin
Ribavirin
Short Answer
 1,2,4-triazole
1,2,4-triazole
Learning Materials
Features
Discover
Chapter 15: Q15-38E (page 477)
Ribavirin, an antiviral agent used against hepatitis C and viral pneumonia, contains a 1,2,4-triazole ring. Why is the ring aromatic?
 Ribavirin
Ribavirin
 1,2,4-triazole
1,2,4-triazole
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw an energy diagram for the three molecular orbitals of the cyclopropenyl system(C3H3). How are these three molecular orbitals occupied in the cyclopropenyl anion, cation, and radical? Which of the three substances is aromatic according to Hückel’s rule?
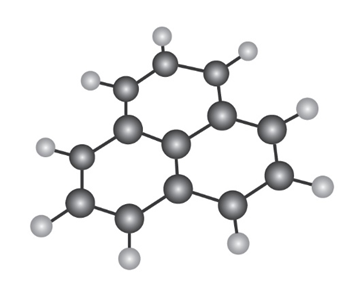
The following molecular model is that of a carbocation. Draw two resonance structures for the carbocation, indicating the positions of the double bonds.
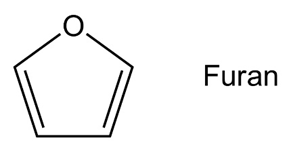
Draw an orbital picture of furan to show how the molecule is aromatic.
Draw structures corresponding to the following names:
(a) 3-Methyl-1,2-benzenediamine
(b) 1,3,5-Benzenetriol
(c) 3-Methyl-2-phenylhexane
(d) o-Aminobenzoic acid
(e) m-Bromophenol
(f) 2,4,6-Trinitrophenol (picric acid)
Propose structures for compounds that fit the following descriptions:
(a) NMR: (4 H, broad singlet);(4 H, quartet, J = 7 Hz); (6 H, triplet, J =7 Hz) IR:.
(b)NMR:(4 H, broad singlet);(1 H, septet, J = 8 Hz);(3 H, singlet);(6 H, doublet, J = 8 Hz) IR:.
What do you think about this solution?
We value your feedback to improve our textbook solutions.