Chapter 15: Q15-15-27 (page 477)
Look at the five resonance structures for phenanthrene (Problem 15-26), and predict which of its carbon-carbon bonds is the shortest.
Short Answer
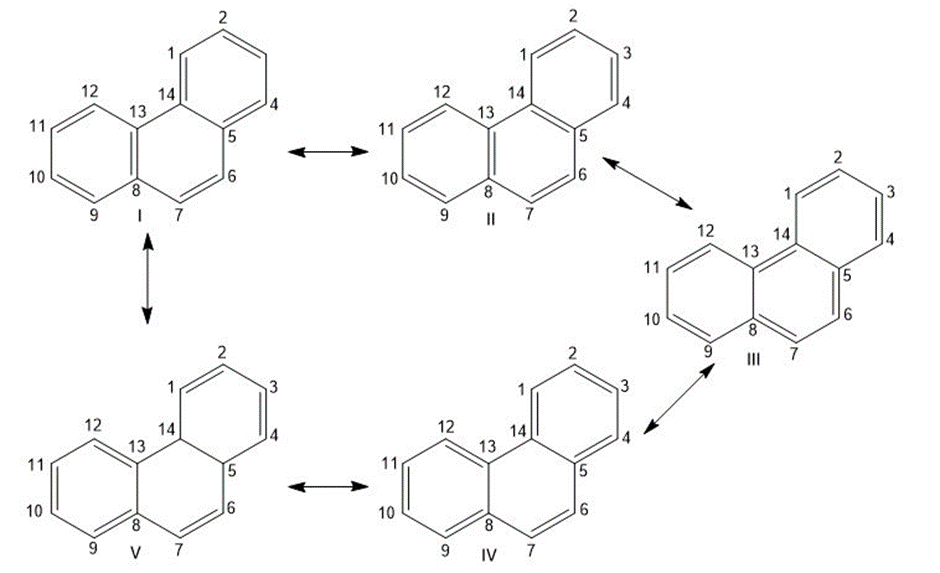
Shorter bond at 6 and 7 carbon
Learning Materials
Features
Discover
Chapter 15: Q15-15-27 (page 477)
Look at the five resonance structures for phenanthrene (Problem 15-26), and predict which of its carbon-carbon bonds is the shortest.

Shorter bond at 6 and 7 carbon
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw structures corresponding to the following names:
(a) 3-Methyl-1,2-benzenediamine
(b) 1,3,5-Benzenetriol
(c) 3-Methyl-2-phenylhexane
(d) o-Aminobenzoic acid
(e) m-Bromophenol
(f) 2,4,6-Trinitrophenol (picric acid)
Thiamin, or vitamin B1, contains a positively charged five-membered nitrogen–sulfur heterocycle called a thiazolium ring. Explain why the thiazolium ring is aromatic.
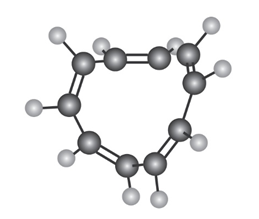
All-cis cyclodecapentaene is a stable molecule that shows a single absorption in its NMR spectrum at . Tell whether it is aromatic, and explain its NMR spectrum.
Look at the three resonance structures of naphthalene shown in Section 15-6, and account for the fact that not all carbon-carbon bonds have the same length. The C1–C2 bond is 136 pm long, whereas the C2–C3 bond is 139 pm long
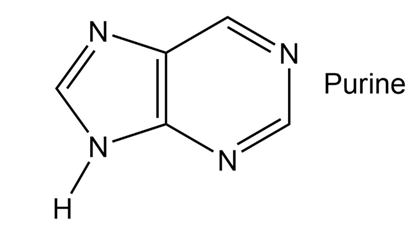
How many electrons does each of the four nitrogen atoms in purine contribute to the aromatic system?
What do you think about this solution?
We value your feedback to improve our textbook solutions.