Chapter 15: Q15-12P (page 469)
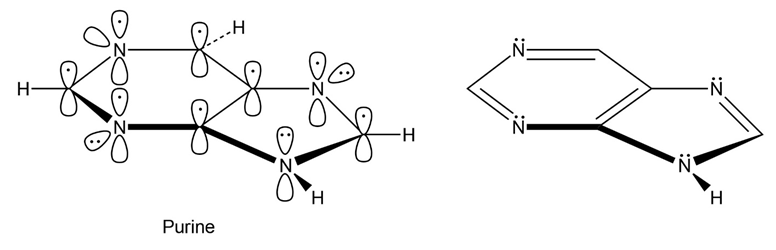
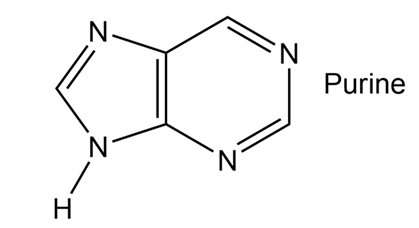
How many electrons does each of the four nitrogen atoms in purine contribute to the aromatic system?
Short Answer
A total of five electrons are donated by the nitrogen in the ring.
The NH shown donate 1 lone pair that 2 electrons which take part in delocalization and other three donate 1 electron.