Chapter 15: Q15-13E (page 477)
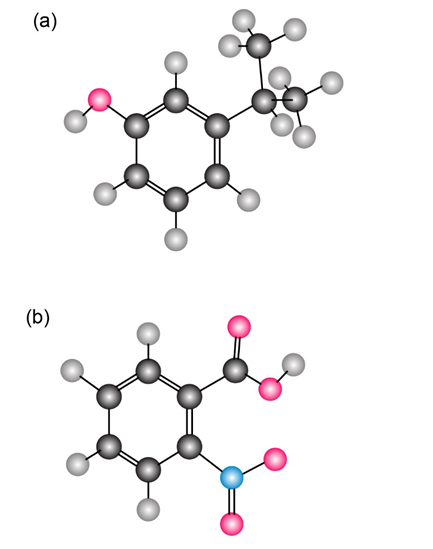
Give IUPAC names for the following substances (red 5 O, blue 5 N):
Short Answer
- m-isopropyl phenol
- o-nitrobenzoic acid
Learning Materials
Features
Discover
Chapter 15: Q15-13E (page 477)
Give IUPAC names for the following substances (red 5 O, blue 5 N):

All the tools & learning materials you need for study success - in one app.
Get started for free
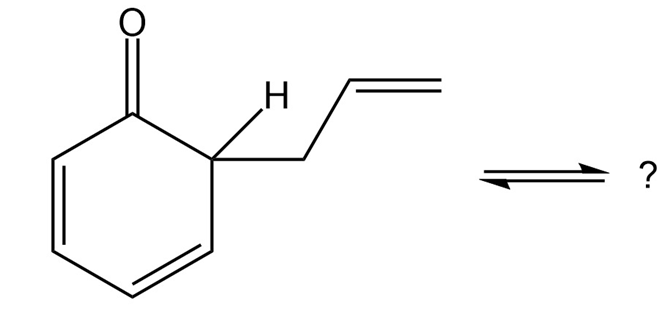
The compound below is the product initially formed in a Claisen rearrangement (Section 18-4). This product is not isolated but tautomerizes to its enol form. Give the structure of the enol and provide an explanation as to why the enol tautomer is favored.
Ribavirin, an antiviral agent used against hepatitis C and viral pneumonia, contains a 1,2,4-triazole ring. Why is the ring aromatic?
 Ribavirin
Ribavirin
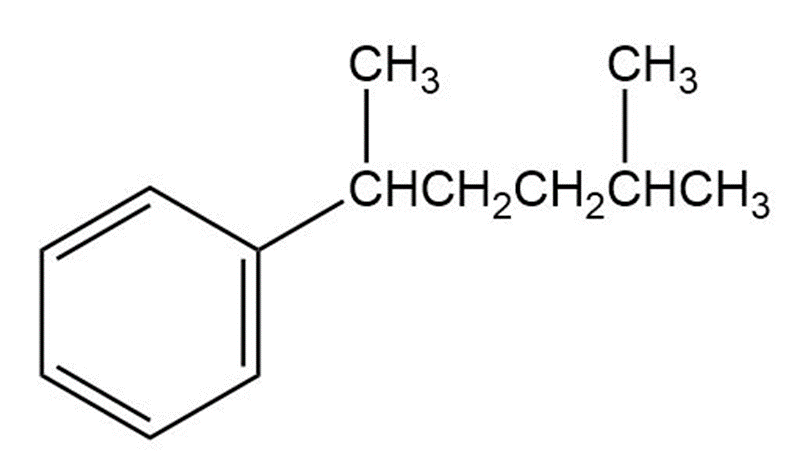
Give IUPAC names for the following compounds
a)
Draw structures corresponding to the following IUPAC names:
(a) p-Bromochlorobenzene
(b) p-Bromotoluene
(c) m-Chloroaniline
(d) 1-Chloro-3,5-dimethylbenzene
The substitution reaction of toluene with Br2 can, in principle, lead to the formation of three isomeric Bromo toluene products. In practice, however, only o- and p-Bromo toluene are formed in substantial amounts. The meta isomer is not formed. Draw the structures of the three possible carbocation intermediates (Problem 15-51), and explain why ortho and para products predominate over meta products.
What do you think about this solution?
We value your feedback to improve our textbook solutions.