Chapter 15: Q 15-15-18 E-a (page 477)
Give IUPAC names for the following compounds
a)
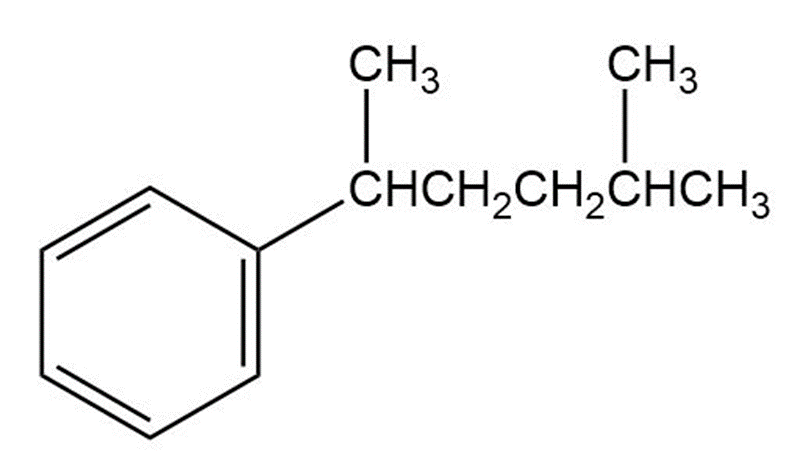
Short Answer
a)
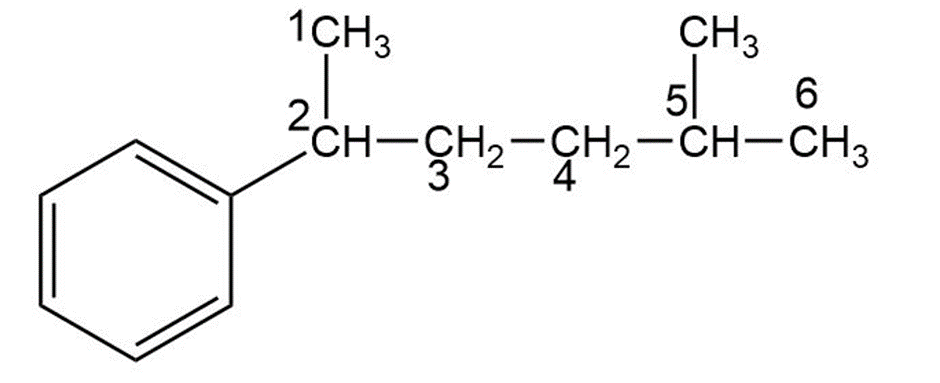
5-methyl-2-phenylhexane
Learning Materials
Features
Discover
Chapter 15: Q 15-15-18 E-a (page 477)
Give IUPAC names for the following compounds
a)

a)
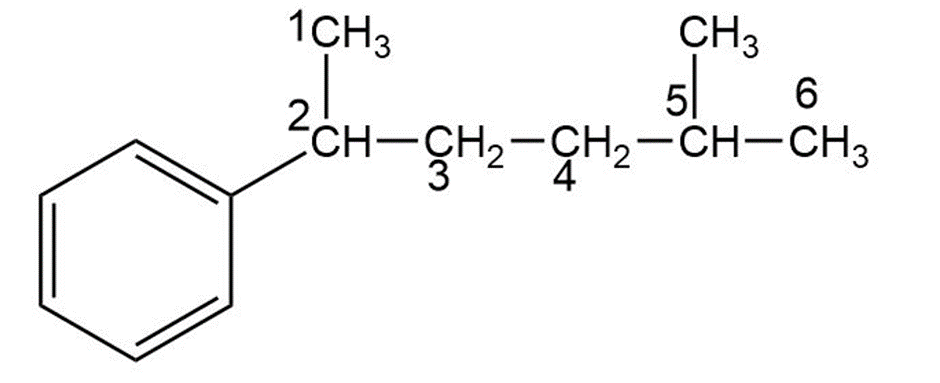
5-methyl-2-phenylhexane
All the tools & learning materials you need for study success - in one app.
Get started for free
Give IUPAC names for the following compounds
d)
Cycloheptatrienone is stable, but cyclopentadienone is so reactive that it can’t be isolated. Explain, taking the polarity of the carbonyl group into account.
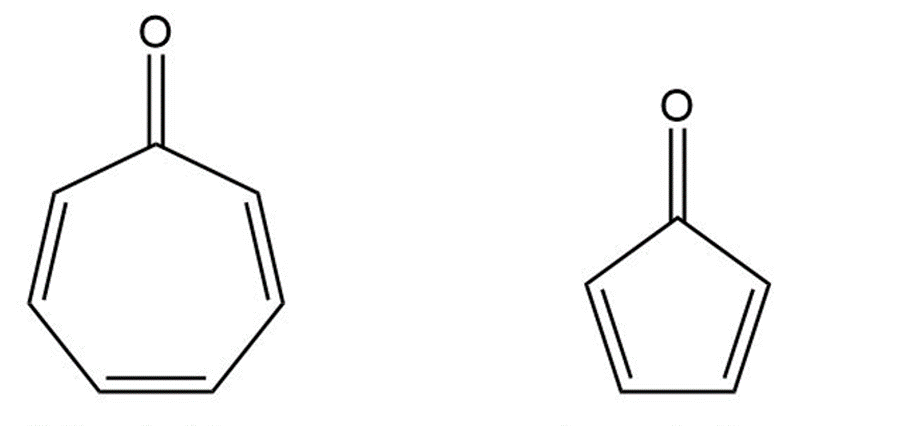
Cycloheptatrienone Cyclopentadienone
To be aromatic, a molecule must have 4n+2 p electrons and must have a planar, monocyclic system of conjugation. Cyclodecapentaene fulfills one of these criteria but not the other and has resisted all attempts at synthesis. Explain.
Draw an energy diagram for the three molecular orbitals of the cyclopropenyl system(C3H3). How are these three molecular orbitals occupied in the cyclopropenyl anion, cation, and radical? Which of the three substances is aromatic according to Hückel’s rule?
Look at the five resonance structures for phenanthrene (Problem 15-26), and predict which of its carbon-carbon bonds is the shortest.
What do you think about this solution?
We value your feedback to improve our textbook solutions.