Chapter 15: Q 15-15-18 E-d (page 477)
Give IUPAC names for the following compounds
d)
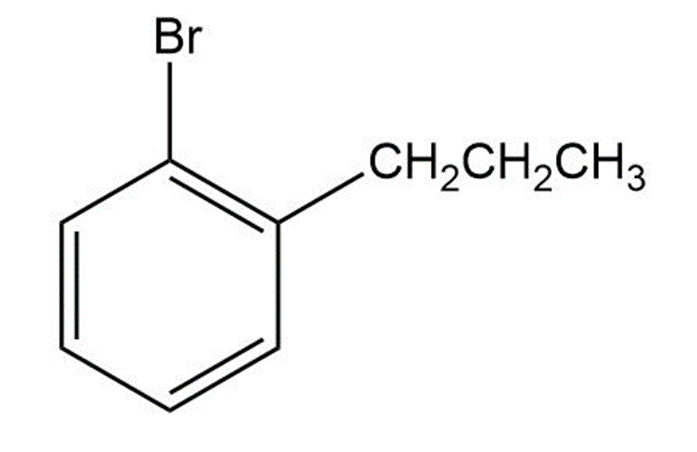
Short Answer
d)
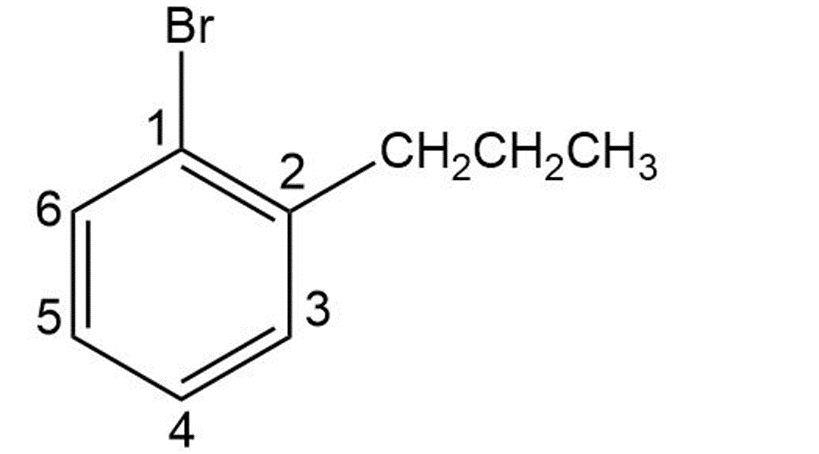
1-bromo-2-propylbenzene
Learning Materials
Features
Discover
Chapter 15: Q 15-15-18 E-d (page 477)
Give IUPAC names for the following compounds
d)

d)

1-bromo-2-propylbenzene
All the tools & learning materials you need for study success - in one app.
Get started for free
Give IUPAC names for the following compounds
c)
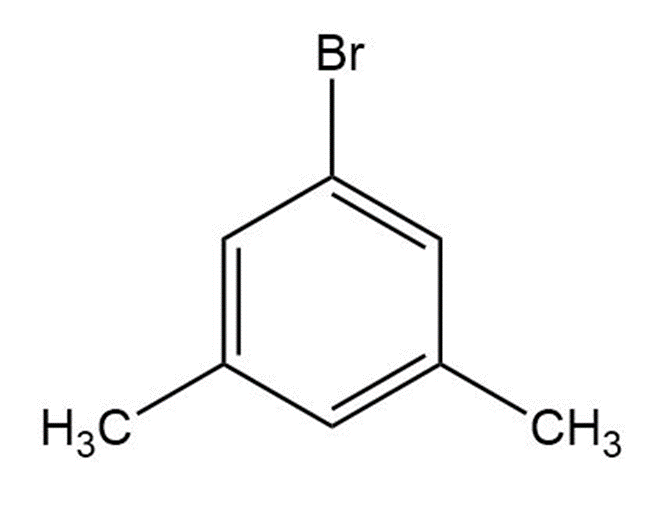
Draw structures corresponding to the following names:
(a) 3-Methyl-1,2-benzenediamine
(b) 1,3,5-Benzenetriol
(c) 3-Methyl-2-phenylhexane
(d) o-Aminobenzoic acid
(e) m-Bromophenol
(f) 2,4,6-Trinitrophenol (picric acid)
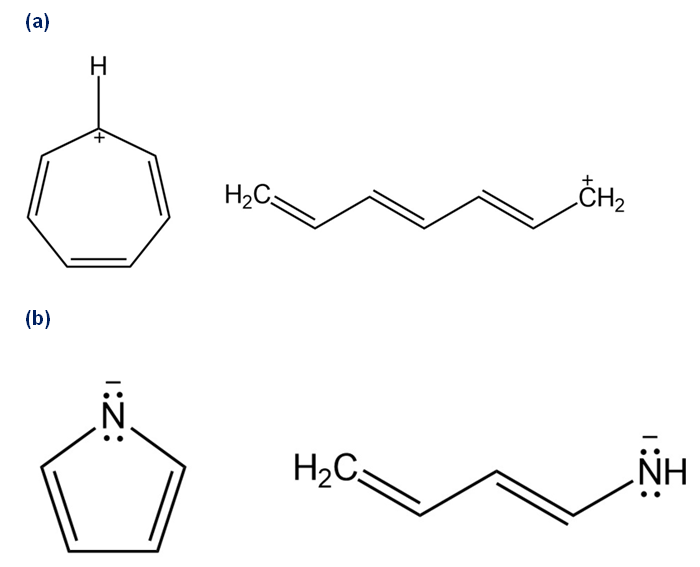
Draw all of the resonance forms for each. What patterns emerge?
The substitution reaction of toluene with Br2 can, in principle, lead to the formation of three isomeric Bromo toluene products. In practice, however, only o- and p-Bromo toluene are formed in substantial amounts. The meta isomer is not formed. Draw the structures of the three possible carbocation intermediates (Problem 15-51), and explain why ortho and para products predominate over meta products.
Draw structures corresponding to the following names:
(a) 3-Methyl-1,2-benzenediamine
(b) 1,3,5-Benzenetriol
(c) 3-Methyl-2-phenylhexane
(d) o-Aminobenzoic acid
(e) m-Bromophenol
(f) 2,4,6-Trinitrophenol (picric acid)
What do you think about this solution?
We value your feedback to improve our textbook solutions.