Chapter 15: Q15-53E (page 477)
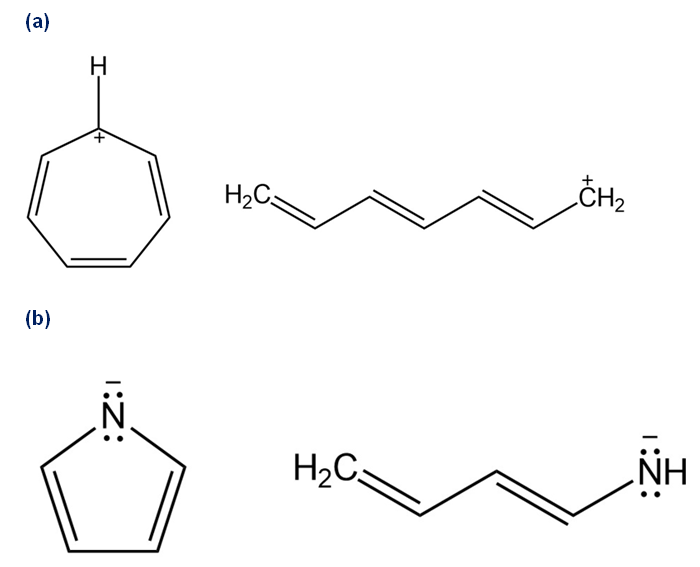
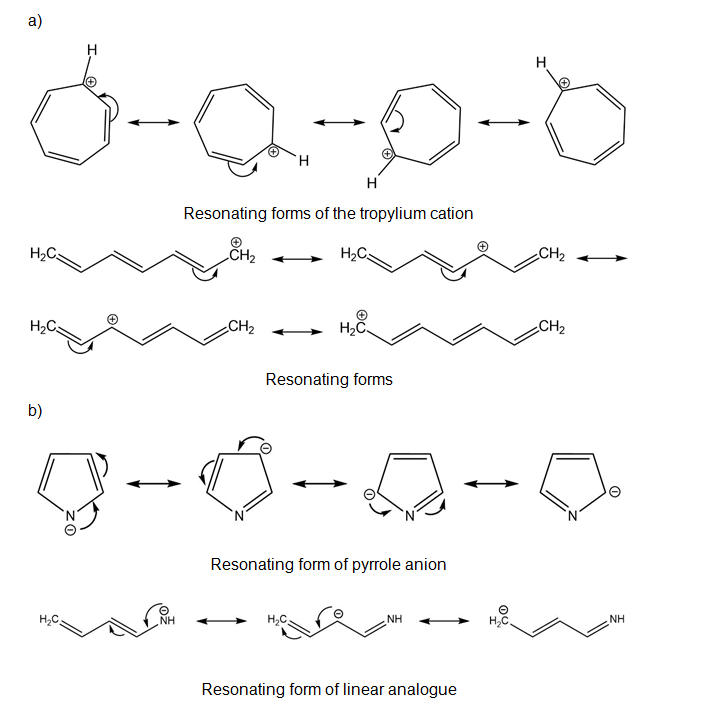
Draw all of the resonance forms for each. What patterns emerge?
Short Answer
Learning Materials
Features
Discover
Chapter 15: Q15-53E (page 477)
Draw all of the resonance forms for each. What patterns emerge?


All the tools & learning materials you need for study success - in one app.
Get started for free
Propose structures for aromatic hydrocarbons that meet the following descriptions:
(a) C9H12; gives only one C9H11Brproduct on substitution of hydrogen on the aromatic ring with bromine
(b)C10 H14; gives only one C10H13Clproduct on substitution of hydrogen on the aromatic ring with chlorine
(c) C8H10; gives three C8H9Brproducts on substitution of hydrogen on the aromatic ring with bromine
(d)C10 H14 ; gives two C10H13Clproducts on substitution of hydrogen on the aromatic ring with chlorine
Calicene, like azulene (Problem 15-17), has an unusually large dipole moment for a hydrocarbon. Explain, using resonance structures.
 Calicene
Calicene
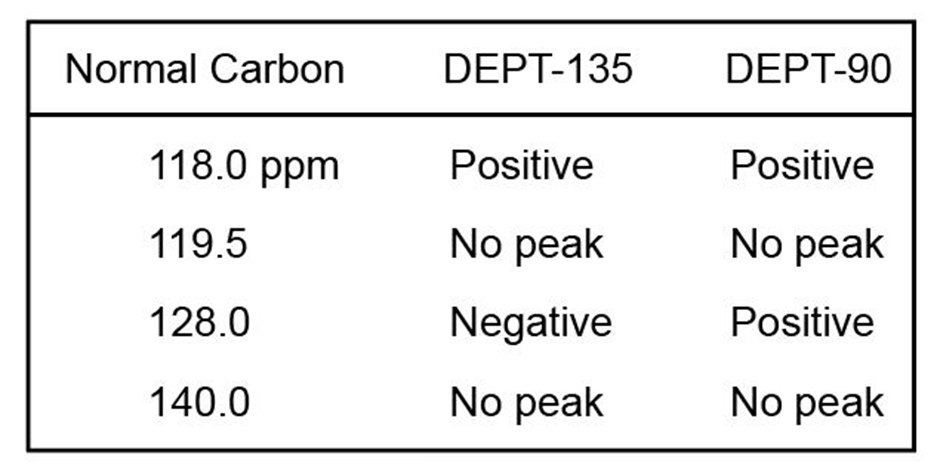
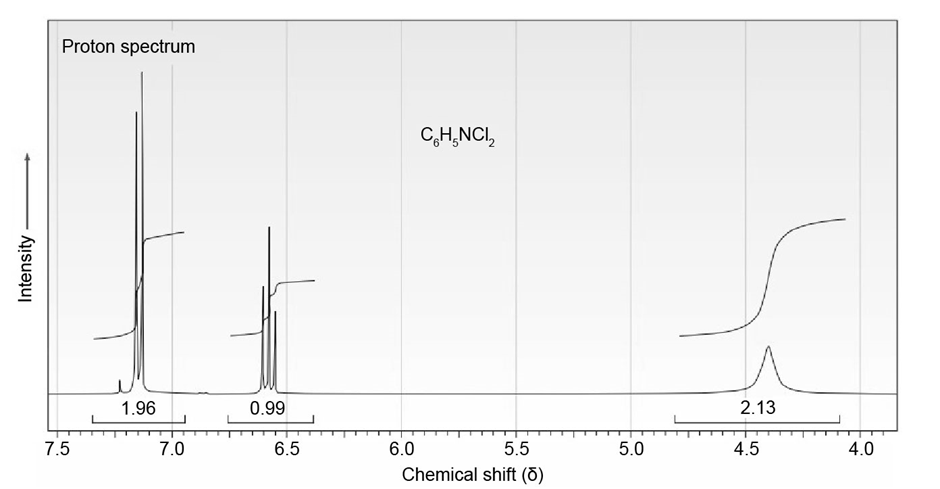
The proton NMR spectrum of a compound with formula is shown. The normal carbon-13 and DEPT experimental results are tabulated. The infrared spectrum shows peaks at 3432 and and a series of medium-sized peaks between 1618 and . Draw the structure of this compound.
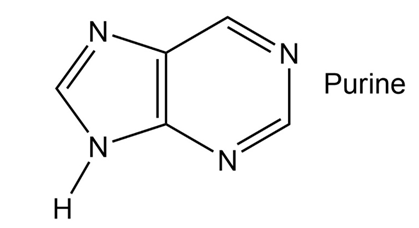
How many electrons does each of the four nitrogen atoms in purine contribute to the aromatic system?
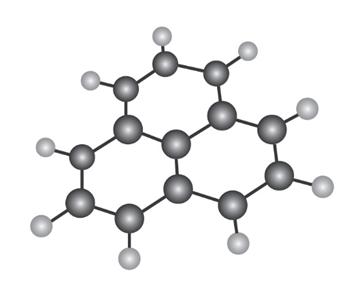
The following molecular model is that of a carbocation. Draw two resonance structures for the carbocation, indicating the positions of the double bonds.
What do you think about this solution?
We value your feedback to improve our textbook solutions.