Chapter 15: Q15-35E (page 477)
Calicene, like azulene (Problem 15-17), has an unusually large dipole moment for a hydrocarbon. Explain, using resonance structures.
 Calicene
Calicene
Short Answer
 Dipole moment in calicene
Dipole moment in calicene
 Resonance contributors calicene
Resonance contributors calicene
Learning Materials
Features
Discover
Chapter 15: Q15-35E (page 477)
Calicene, like azulene (Problem 15-17), has an unusually large dipole moment for a hydrocarbon. Explain, using resonance structures.
 Calicene
Calicene
 Dipole moment in calicene
Dipole moment in calicene
 Resonance contributors calicene
Resonance contributors calicene
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw structures corresponding to the following names:
(a) 3-Methyl-1,2-benzenediamine
(b) 1,3,5-Benzenetriol
(c) 3-Methyl-2-phenylhexane
(d) o-Aminobenzoic acid
(e) m-Bromophenol
(f) 2,4,6-Trinitrophenol (picric acid)
Propose a structure for a molecule that has the following NMR spectrum and has IR absorptions at 700, 740, and :
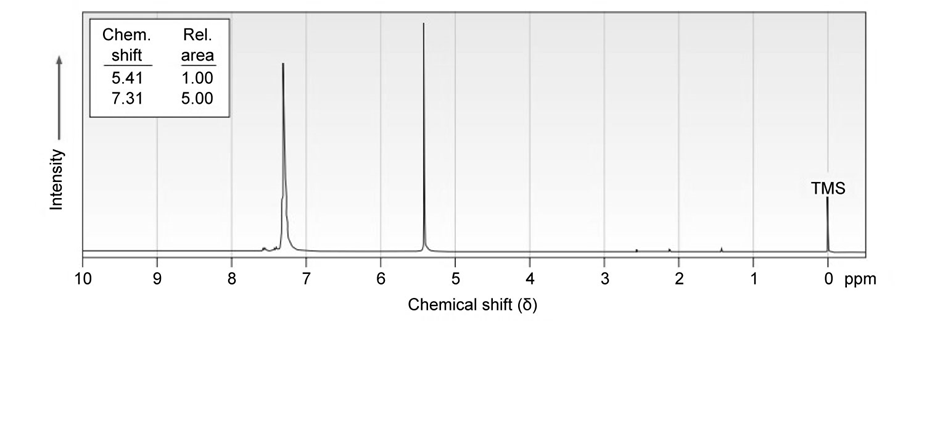
Aromatic substitution reactions occur by the addition of an electrophile such as to an aromatic ring to yield an allylic carbocation intermediate, followed by loss of . Show the structure of the intermediate formed by the reaction of benzene with .
Look at the five resonance structures for phenanthrene (Problem 15-26), and predict which of its carbon-carbon bonds is the shortest.
Give IUPAC names for the following compounds
c)
What do you think about this solution?
We value your feedback to improve our textbook solutions.