Chapter 15: Q48E (page 477)
Propose a structure for a molecule that has the following NMR spectrum and has IR absorptions at 700, 740, and :
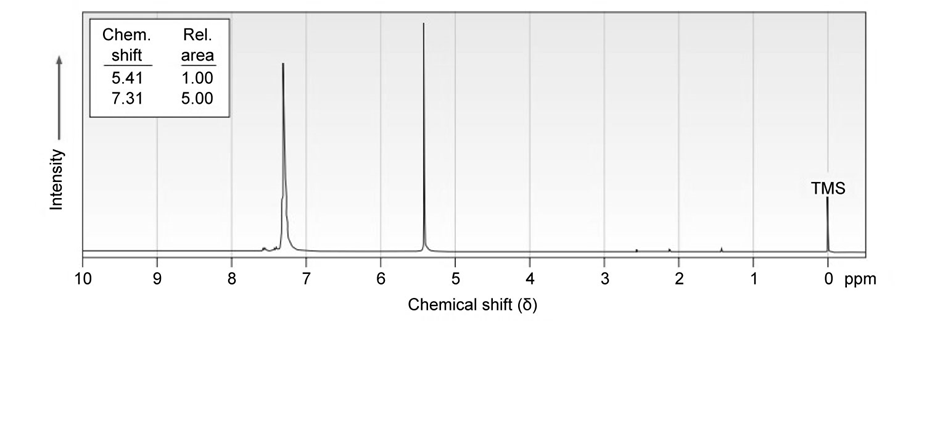
Short Answer
 Formation of the given structure
Formation of the given structure
Learning Materials
Features
Discover
Chapter 15: Q48E (page 477)
Propose a structure for a molecule that has the following NMR spectrum and has IR absorptions at 700, 740, and :

 Formation of the given structure
Formation of the given structure
All the tools & learning materials you need for study success - in one app.
Get started for free
Give IUPAC names for the following compounds
a)
Draw the five resonance structures of the cyclopentadienyl anion. are all carbon-carbon bonds equivalent? How many absorption lines would you expect to see in the NMR and NMR spectra of the anion?
Bextra, a COX-2 inhibitor once used in the treatment of arthritis, contains an isoxazole ring. Why is the ring aromatic?
 Bextra
Bextra
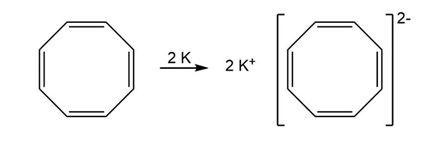
Cyclooctatetraene readily reacts with potassium metal to form the stable cyclooctatetraene dianion, . Why do you suppose this reaction occurs so easily? What geometry do you expect for the cyclooctatetraene dianion?
Ribavirin, an antiviral agent used against hepatitis C and viral pneumonia, contains a 1,2,4-triazole ring. Why is the ring aromatic?
 Ribavirin
Ribavirin
What do you think about this solution?
We value your feedback to improve our textbook solutions.