Chapter 15: Q43E (page 477)
Bextra, a COX-2 inhibitor once used in the treatment of arthritis, contains an isoxazole ring. Why is the ring aromatic?
 Bextra
Bextra
Short Answer
Isoxazole ring
(aromatic)
Learning Materials
Features
Discover
Chapter 15: Q43E (page 477)
Bextra, a COX-2 inhibitor once used in the treatment of arthritis, contains an isoxazole ring. Why is the ring aromatic?
 Bextra
Bextra

Isoxazole ring
(aromatic)
All the tools & learning materials you need for study success - in one app.
Get started for free
All-cis cyclodecapentaene is a stable molecule that shows a single absorption in its NMR spectrum at . Tell whether it is aromatic, and explain its NMR spectrum.
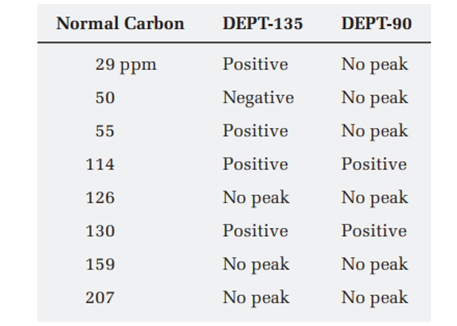
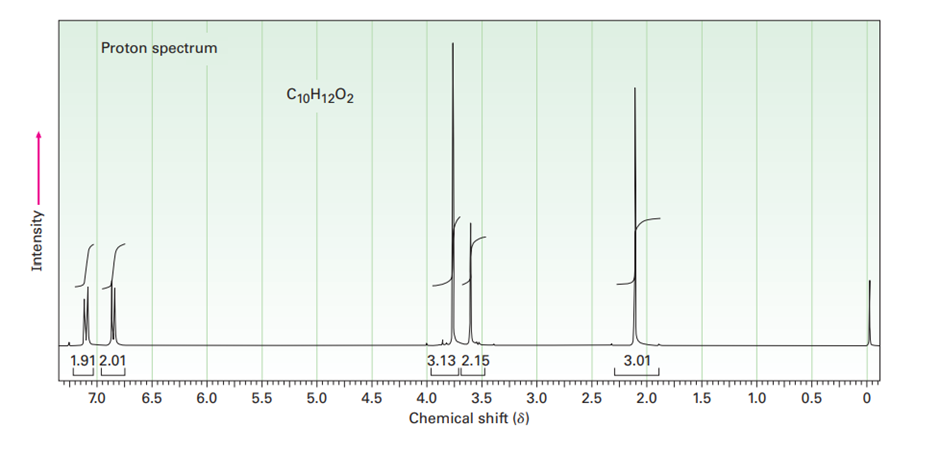
The proton NMR spectrum for a compound with formula is shown below. The infrared spectrum has a strong band at . The normal carbon-13 NMR spectral results are tabulated along with the DEPT-135 and DEPT-90 information. Draw the structure of this compound.
Cyclopropane is highly reactive because of its large amount of angle strain. Methylcyclopropenone, although even more strained than cyclopropanone, is nevertheless quite stable and can even be distilled. Explain, taking the polarity of the carbonyl group into account.
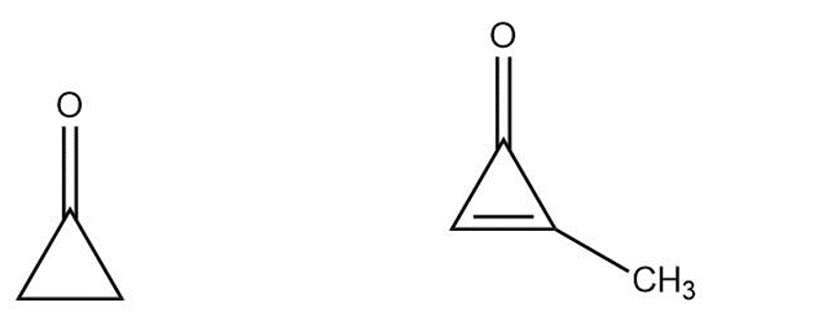
Cyclopropanone Methylcyclopropenone
Propose structures for compounds that fit the following descriptions:
(a) NMR: (4 H, broad singlet);(4 H, quartet, J = 7 Hz); (6 H, triplet, J =7 Hz) IR:.
(b)NMR:(4 H, broad singlet);(1 H, septet, J = 8 Hz);(3 H, singlet);(6 H, doublet, J = 8 Hz) IR:.
Draw and name all possible isomers of the following:
What do you think about this solution?
We value your feedback to improve our textbook solutions.