Chapter 15: Q42E (page 477)
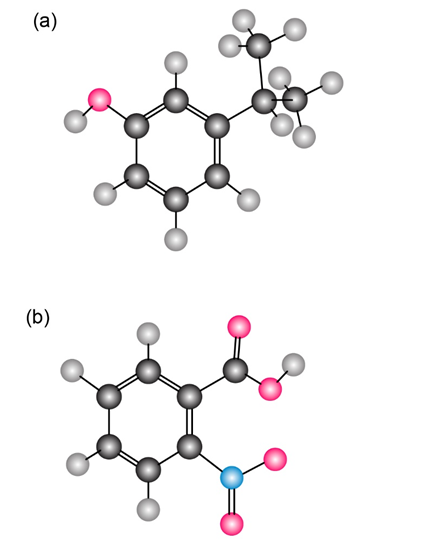
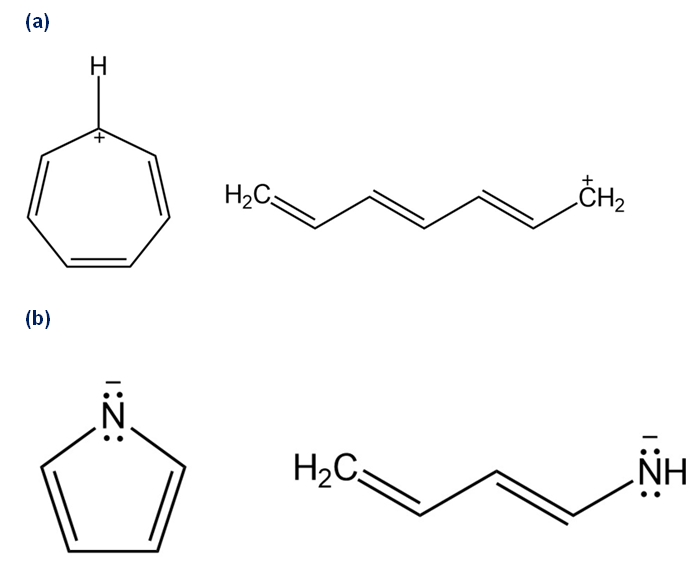
On reaction with acid, 4-pyrone is protonated on the carbonyl-group oxygen to give a stable cationic product. Using resonance structures and the Hückel’s rule, explain why the protonated product is so stable.
 4-Pyrone
4-Pyrone
Short Answer
 Identical resonance forms
Identical resonance forms
 Identical resonance forms
Identical resonance forms

 Indole
Indole