Chapter 15: Q15-34E (page 477)
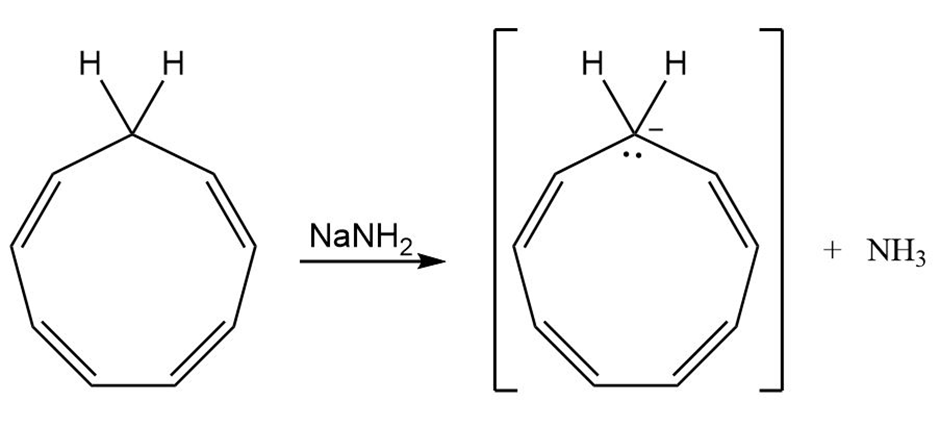
How might you convert 1, 3, 5, 7-cyclononatetraene to an aromatic substance?
Short Answer
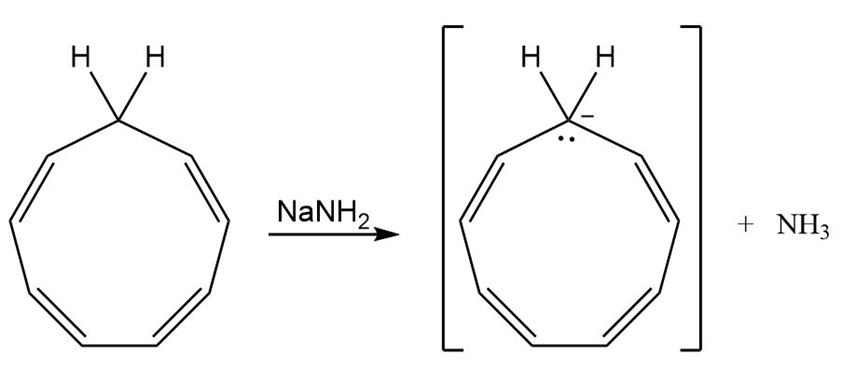
1, 3, 5, 7-cyclononatetraene cyclononatetraenyl anion
(8 electrons) (10 electrons)
Conversion of 1, 3, 5, 7-cyclononatetraene to aromatic substance