Chapter 15: Q15-15-25E (page 477)
Anthracene has four resonance structures, one of which is shown. Draw the other three
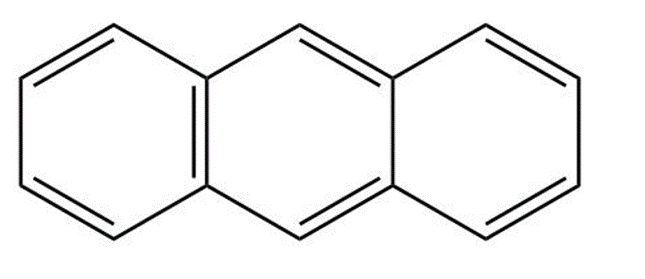
Anthracene
Short Answer
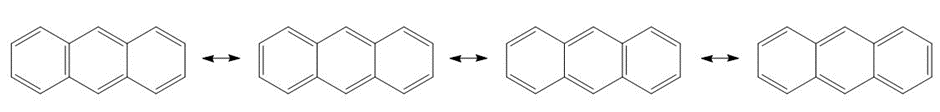
Four resonance structures of anthracene
Learning Materials
Features
Discover
Chapter 15: Q15-15-25E (page 477)
Anthracene has four resonance structures, one of which is shown. Draw the other three

Anthracene

Four resonance structures of anthracene
All the tools & learning materials you need for study success - in one app.
Get started for free
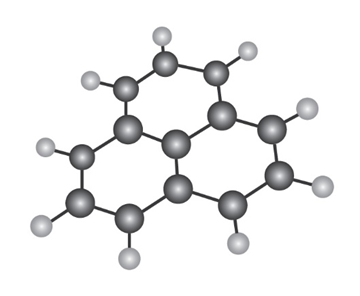
The following molecular model is that of a carbocation. Draw two resonance structures for the carbocation, indicating the positions of the double bonds.
Propose structures for compounds that fit the following descriptions:
(a) NMR: (4 H, broad singlet);(4 H, quartet, J = 7 Hz); (6 H, triplet, J =7 Hz) IR:.
(b)NMR:(4 H, broad singlet);(1 H, septet, J = 8 Hz);(3 H, singlet);(6 H, doublet, J = 8 Hz) IR:.
Look at the five resonance structures for phenanthrene (Problem 15-26), and predict which of its carbon-carbon bonds is the shortest.
All-cis cyclodecapentaene is a stable molecule that shows a single absorption in its NMR spectrum at . Tell whether it is aromatic, and explain its NMR spectrum.
7 Propose structures for aromatic compounds that have the following NMR spectra:
(a)
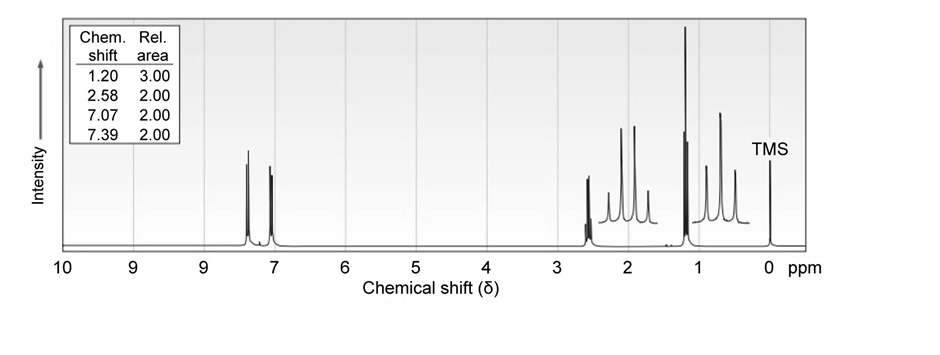
b)
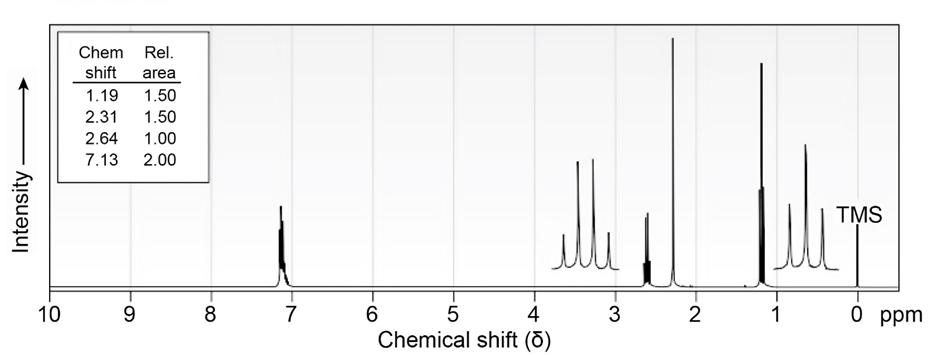
c)
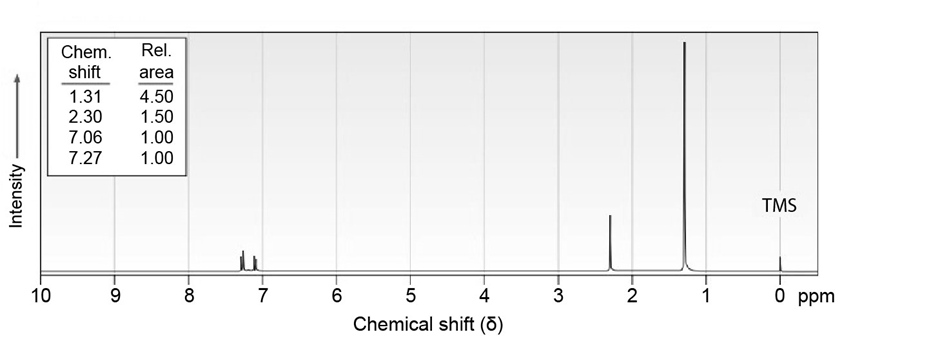
What do you think about this solution?
We value your feedback to improve our textbook solutions.