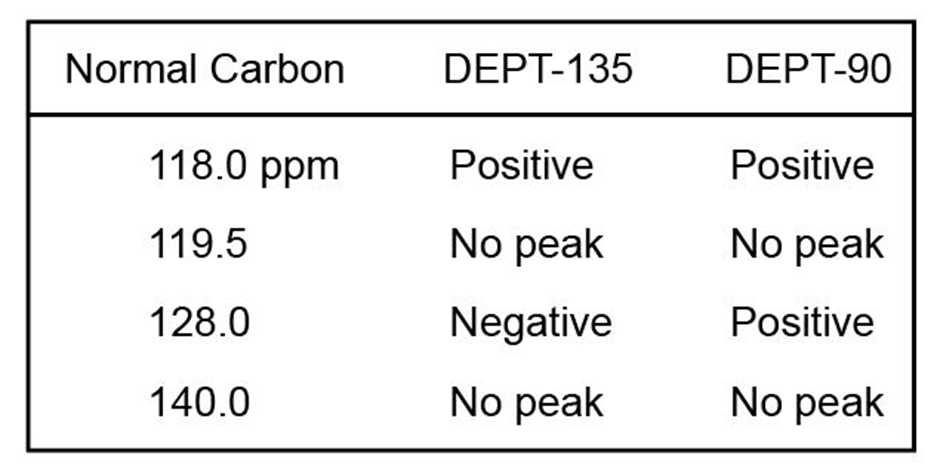
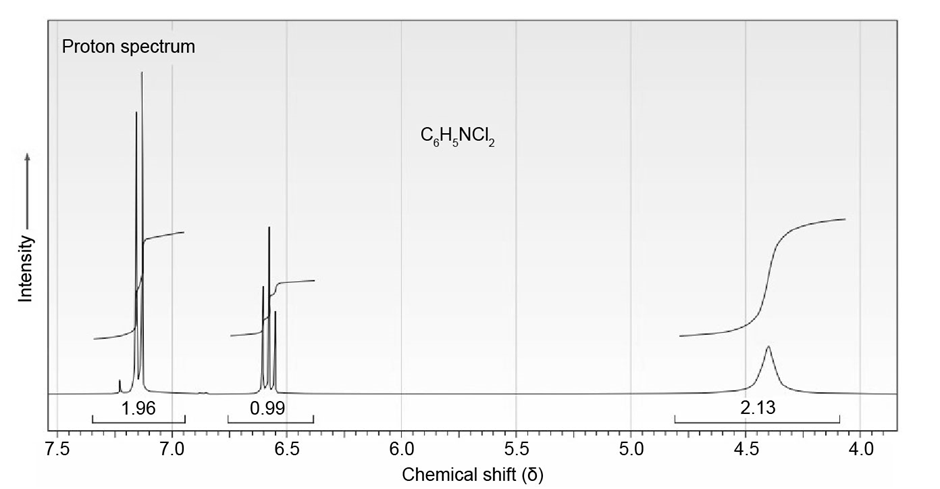
Chapter 15: Q15-15-32E (page 477)
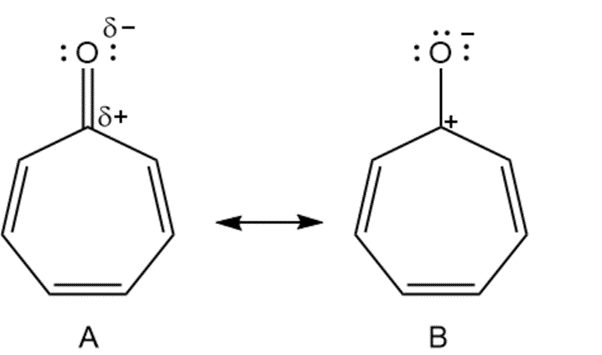
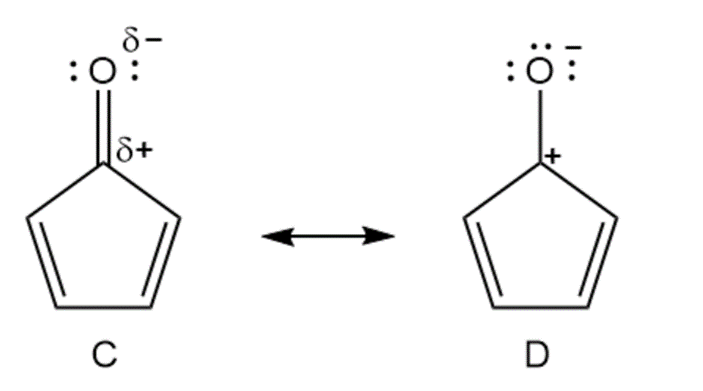
Cycloheptatrienone is stable, but cyclopentadienone is so reactive that it can’t be isolated. Explain, taking the polarity of the carbonyl group into account.
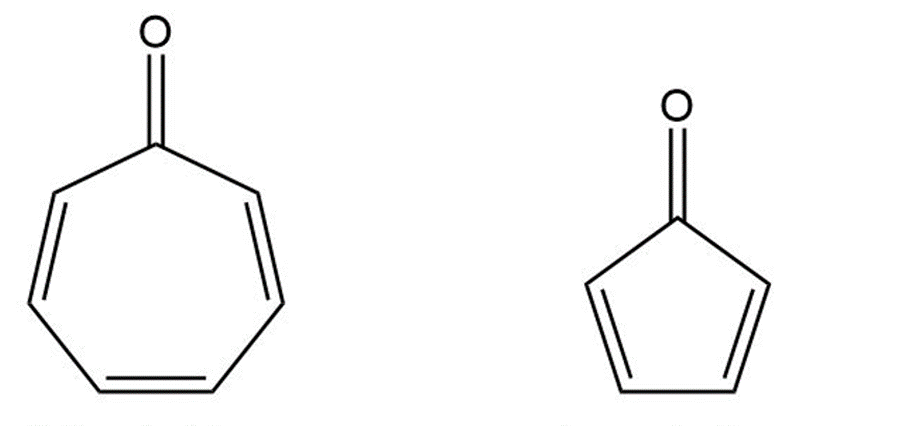
Cycloheptatrienone Cyclopentadienone
Short Answer
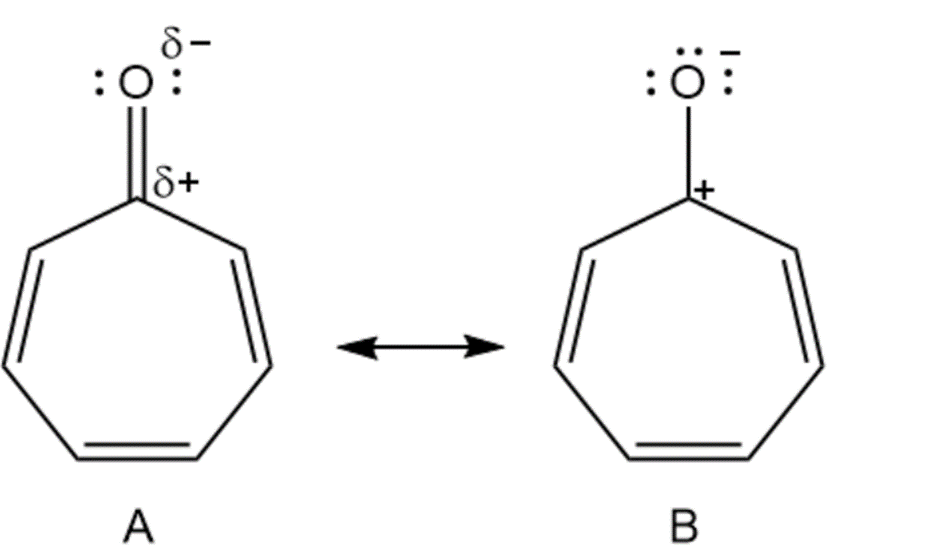
Cycloheptatrienone
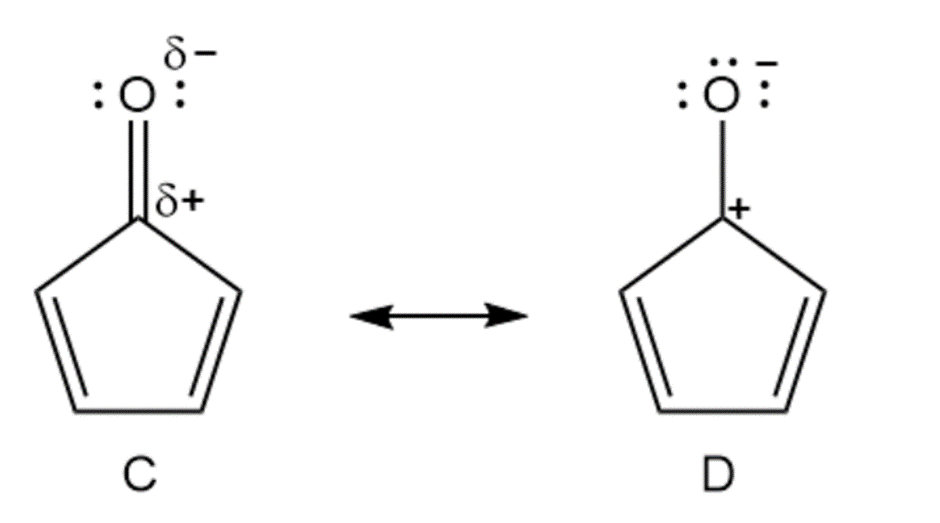
Cyclopentadienone


 Methyl orange
Methyl orange C.I. Acid Red 74
C.I. Acid Red 74