Chapter 15: Q15-15-33E (page 477)
Which would you expect to be most stable, Cyclononatetraenyl radical, cation, or anion?
Short Answer
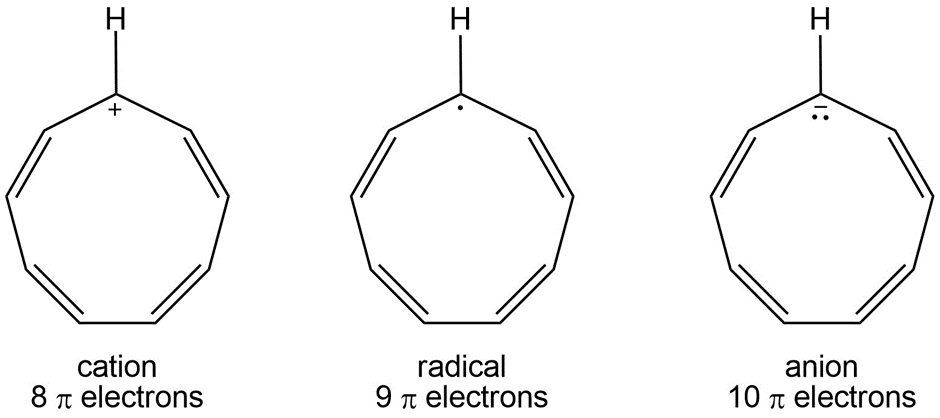
Cyclononatetraenyl cation Cyclononatetraenyl radical, Cyclononatetraenyl
(Most stable) anion
Learning Materials
Features
Discover
Chapter 15: Q15-15-33E (page 477)
Which would you expect to be most stable, Cyclononatetraenyl radical, cation, or anion?

Cyclononatetraenyl cation Cyclononatetraenyl radical, Cyclononatetraenyl
(Most stable) anion
All the tools & learning materials you need for study success - in one app.
Get started for free
N-Phenylsydnone, so-named because it was first studied at the University of Sydney, Australia, behaves like a typical aromatic molecule. Explain, using the Hückel’s rule .
 N-phenylsydnone
N-phenylsydnone
On reaction with acid, 4-pyrone is protonated on the carbonyl-group oxygen to give a stable cationic product. Using resonance structures and the Hückel’s rule, explain why the protonated product is so stable.
 4-Pyrone
4-Pyrone
Give IUPAC names for the following compounds:
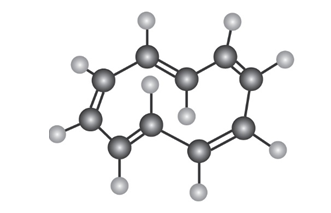
To be aromatic, a molecule must have 4n+2 p electrons and must have a planar, monocyclic system of conjugation. Cyclodecapentaene fulfills one of these criteria but not the other and has resisted all attempts at synthesis. Explain.
The relative energy levels of the five molecular orbitals of the cyclopentadienyl system are similar to those in benzene. That is, there is a single lowest energy MO, above which the orbitals come in degenerate pairs. Draw a diagram like that in Figure 15-5, and tell which of the five orbitals are occupied in the cation, radical, and anion.
What do you think about this solution?
We value your feedback to improve our textbook solutions.