Chapter 15: Q22P (page 477)
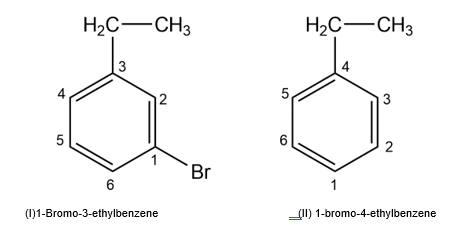
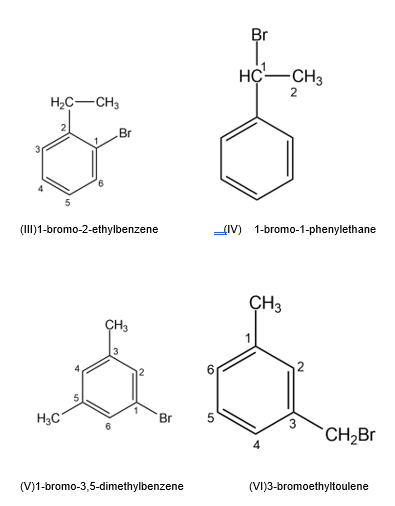
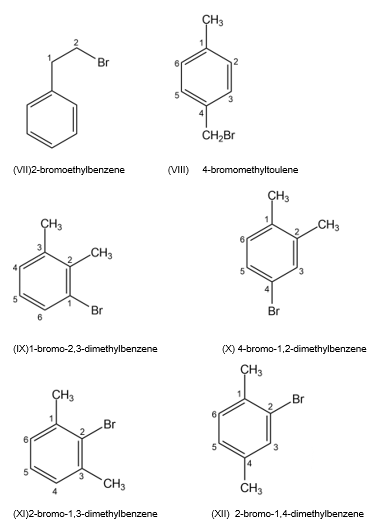
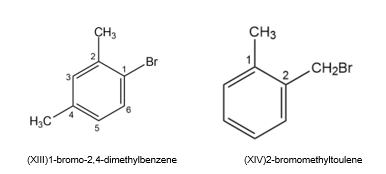
Draw and name all possible aromatic compounds with the formula C8H9Br.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 15: Q22P (page 477)
Draw and name all possible aromatic compounds with the formula C8H9Br.
Answer




All the tools & learning materials you need for study success - in one app.
Get started for free
Look at the five resonance structures for phenanthrene (Problem 15-26), and predict which of its carbon-carbon bonds is the shortest.
Propose structures for compounds that fit the following descriptions:
(a) NMR: (4 H, broad singlet);(4 H, quartet, J = 7 Hz); (6 H, triplet, J =7 Hz) IR:.
(b)NMR:(4 H, broad singlet);(1 H, septet, J = 8 Hz);(3 H, singlet);(6 H, doublet, J = 8 Hz) IR:.
Cyclopropane is highly reactive because of its large amount of angle strain. Methylcyclopropenone, although even more strained than cyclopropanone, is nevertheless quite stable and can even be distilled. Explain, taking the polarity of the carbonyl group into account.
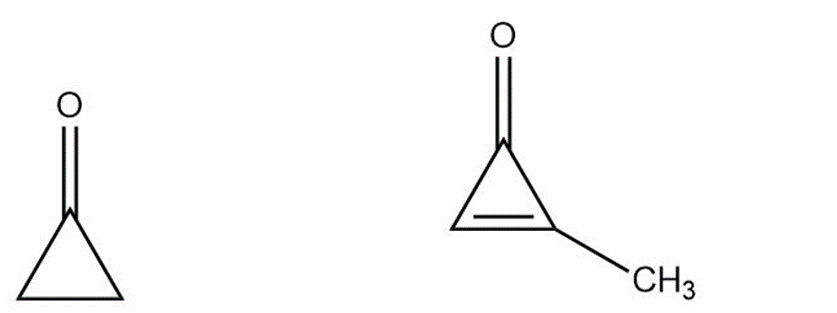
Cyclopropanone Methylcyclopropenone
Pyridine is a flat, hexagonal molecule with bond angles of 120°. It undergoes substitution rather than addition and generally behaves like benzene. Draw a picture of the orbitals of pyridine to explain its properties. Check your answer by looking ahead to Sections 15-5.
Draw structures corresponding to the following names:
(a) 3-Methyl-1,2-benzenediamine
(b) 1,3,5-Benzenetriol
(c) 3-Methyl-2-phenylhexane
(d) o-Aminobenzoic acid
(e) m-Bromophenol
(f) 2,4,6-Trinitrophenol (picric acid)
What do you think about this solution?
We value your feedback to improve our textbook solutions.