Chapter 17: Q11P (page 543)
Use the reaction of a Grignard reagent with a carbonyl compound to synthesize the following compound:
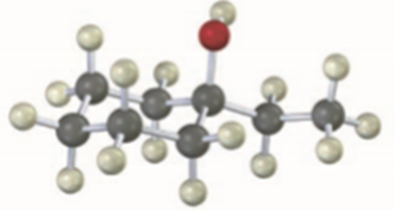
Short Answer
The reaction of cyclohexanone with Grignard reagent is shown as,
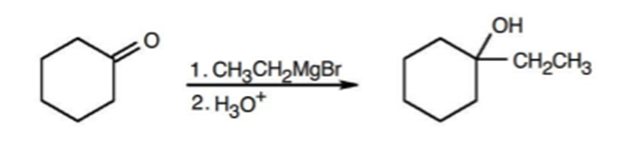
Learning Materials
Features
Discover
Chapter 17: Q11P (page 543)
Use the reaction of a Grignard reagent with a carbonyl compound to synthesize the following compound:

The reaction of cyclohexanone with Grignard reagent is shown as,
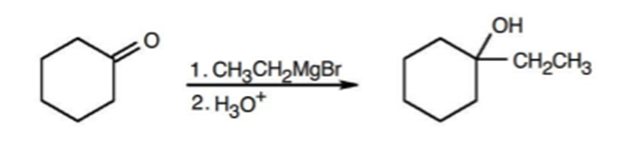
All the tools & learning materials you need for study success - in one app.
Get started for free
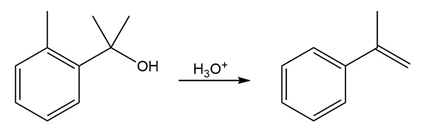
The conversion of 3° alcohols into alkenes under acidic conditions involves two cationic intermediates. For each reaction, draw the complete mechanism using curved arrows.
(a)
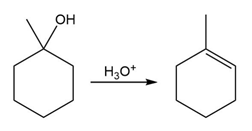
(b)
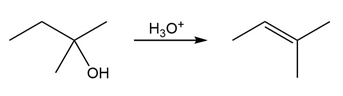
(c)
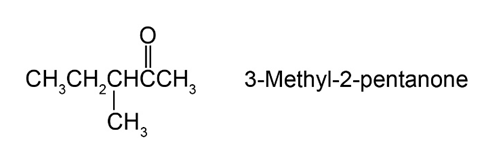
Question:Reaction of (S)-3-methyl-2-pentanone with methyl magnesium bromide followed by acidification yields 2,3-dimethyl-2-pentanol. What is the stereochemistry of the product? Is the product optically active?
Benzoquinone is an excellent dienophile in the Diels – Alder reaction. What product would you expect from reaction of benzoquinone with 1 equivalent of 1,3 butadiene? From reaction with 2 equivalents of 1,3 butadiene?
Predict the product from reaction of the following substance with:
3. ; then
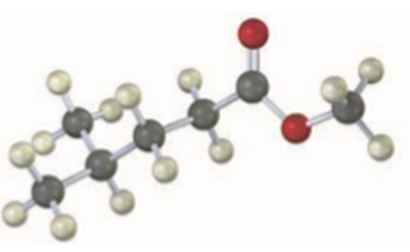
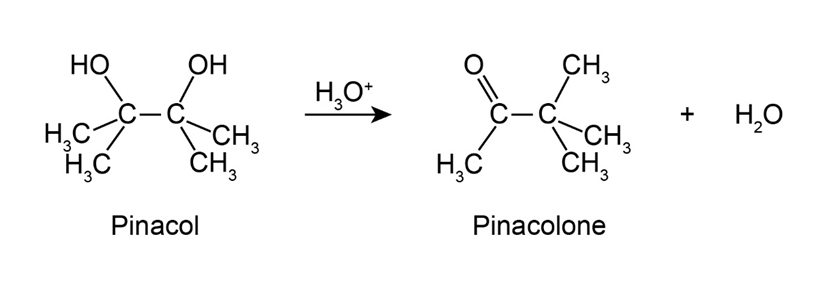
2,3-Dimethyl-2,3-butanediol has the common name pinacol. On heating with aqueous acid, pinacol rearranges to pinacolone, 3,3-dimethyl-2-butanone. Suggest a mechanism for this reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.